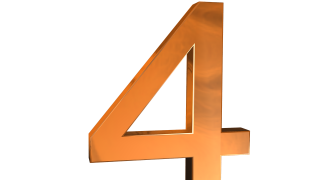India's Dengue Vaccine Candidate Advancing to Approval
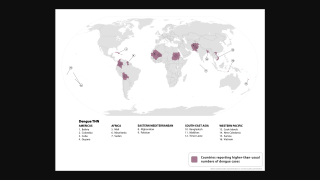
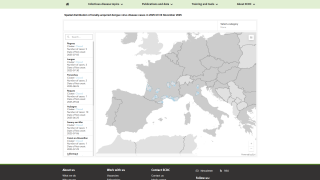
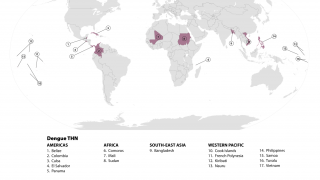
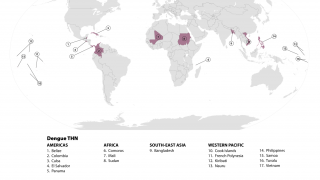
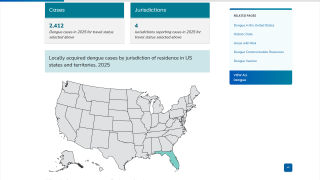
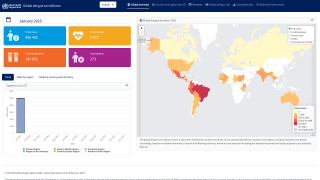
The mosquito-transmitted dengue virus remains a significant public health challenge in tropical regions around the world. In India alone, over one million dengue cases and at least 1,500 deaths have been recorded since 2021.
To address this public health challenge, India's Panacea Biotec Limited has made substantial progress with its vaccine candidate, DengiAll®.
As of March 1, 2026, the company is conducting Phase III clinical trials for DengiAll, a single-dose vaccine designed to provide balanced protection against all four dengue virus serotypes: DENV-1, DENV-2, DENV-3, and DENV-4. This represents a crucial step toward potentially introducing India's first domestically developed dengue vaccine, which would offer an accessible preventive option in areas where dengue is endemic.
"We will try to get this vaccine out there as soon as possible," said Syed Khalid Ali, Chief Scientific Officer at Panacea Biotec, in a media statement on February 26, 2026.
DengiAll is a live-attenuated, recombinant, lyophilized tetravalent vaccine based on attenuated strains licensed from the U.S. National Institutes of Health, which Panacea Biotec has further developed since 2006. Early clinical data from Phase I/II trials in healthy Indian adults showed that a single dose generated strong immunogenicity against all four serotypes, while maintaining a favorable safety profile.
If successful, DengiAll could become a widely available single-dose dengue vaccine, following recent approvals of similar single-shot candidates in other countries, such as Brazil.
Currently, several dengue vaccines are approved in various countries, though availability remains limited globally, with no dengue vaccine licensed for public use in India yet.
Dengvaxia®: The first approved dengue vaccine, a live-attenuated tetravalent chimeric vaccine requiring three doses and pre-vaccination screening for prior dengue infection. It is approved in the U.S. (Puerto Rico). However, production is being discontinued in 2026.
Qdenga®: A live-attenuated tetravalent vaccine administered in two doses, approved without requiring prior dengue testing in most indications. It is authorized in over 40 countries, and over 18 million doses have been distributed.
Butantan-DV: The world's first single-dose tetravalent live-attenuated dengue vaccine, approved by Brazil's Anvisa in November 2025. It is being launched in Brazil, with millions of doses planned for distribution.
Panacea Biotec's initiatives align with global efforts to combat dengue through vaccination, particularly in high-burden regions. Positive outcomes from Phase III trials may lead to regulatory review and potential introduction as soon as next year, depending on favorable data.
While the U.S. Centers for Disease Control and Prevention does not highlight dengue as a health risk when visiting India, as of March 2026, it has recently identified measles and rabies as concerns.
Our Trust Standards: Medical Advisory Committee