Lyme Disease Risk Intensifies in 2026
As tick season ramps up during Spring 2026, health officials urge residents, particularly in the Northeast and Upper Midwest, to stay informed and take proactive steps to reduce exposure.
Public health officials in the United States are projecting a nationwide 15–20% increase in tick populations for 2026. This surge is attributed to favorable environmental conditions, milder winters, and expanding habitats for blacklegged ticks, the primary vectors of Lyme disease.
The peak risk period for Lyme disease transmission is expected to be between May 15 and July 15, 2026, when nymphal ticks—tiny, poppy-seed-sized ticks that are notoriously difficult to detect—are most active. These nymphal ticks are responsible for the majority of human infections because they are hard to spot before they have fed long enough to transmit *Borrelia burgdorferi*, the bacterium that causes Lyme disease.
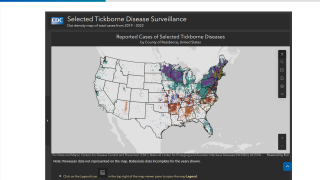
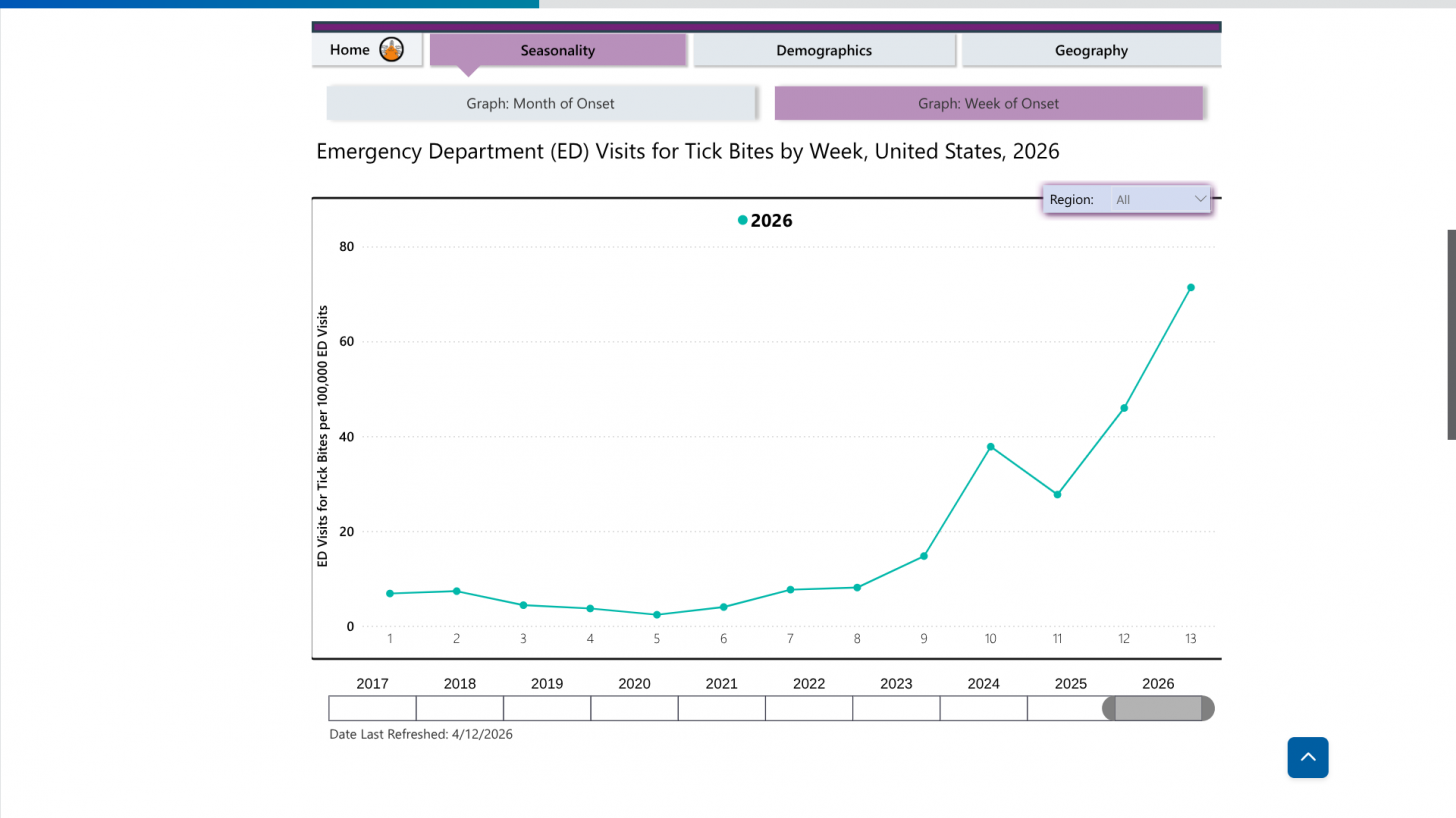
As of April 14, 2026, data from the U.S. Centers for Disease Control and Prevention (CDC) indicate an early increase in medical visits related to tick bites. Emergency department visits for tick bites more than double the typical seasonal rate of about 30 per 100,000 during this time of year.
According to the CDC, this increase follows a record-setting 2025, when national tick-related emergency room visits reached their highest levels since 2019.
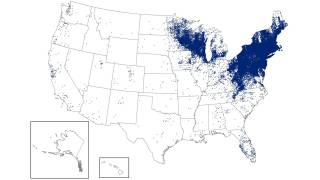
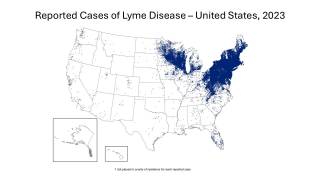
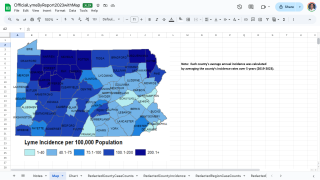
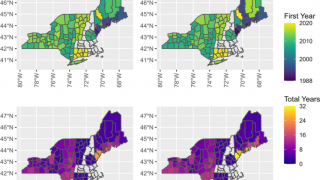
The Northeast region of the U.S. continues to be significantly affected by this issue.
During peak months, the region has recorded between 229 and 283 tick-bite-related visits per 100,000 emergency department visits. Experts anticipate that the states with the highest number of Lyme disease cases in 2026 will again be Pennsylvania, New York, New Jersey, Connecticut, and Massachusetts, along with Minnesota and Wisconsin in the Upper Midwest.
These states have dense blacklegged tick populations and large human populations, which together contribute to the transmission risk.
CDC experts attribute part of the overall rise in reported cases and visits to improved surveillance, more efficient state diagnostic systems, and greater public awareness.
While no Lyme disease vaccine is currently available in the U.S.—the last one was discontinued in 2002—significant progress has been made.
On March 23, 2026, Pfizer and Valneva SE announced positive topline results from their Phase 3 VALOR trial of the investigational vaccine candidate PF-07307405 (LB6V).
The six-valent OspA-based vaccine showed approximately 73% efficacy in preventing confirmed Lyme disease cases in participants aged 5 years and older. The large trial was conducted across high-incidence areas in the U.S., Canada, and Europe.
Pfizer plans to submit the data to the U.S. FDA later this year. If approved, this would mark the first new Lyme disease vaccine available in the U.S. in over two decades.
Our Trust Standards: Medical Advisory Committee