Lyme Disease Test Available At-Home in 51 Countries; No Doctor Required

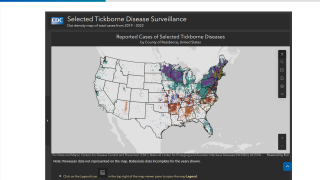
As tick-borne diseases continue to pose health challenges in 2026, many cases of Lyme disease remain undetected, affecting millions of peoplein the United States and Europe.
Traditional testing methods for these diseases often lack sensitivity, resulting in missed or delayed diagnoses.
According to the U.S. Centers for Disease Control and Prevention (CDC), numerous Lyme disease cases go undiagnosed or underreported. This occurs because the characteristic rash does not always appear, symptoms may resemble those of other illnesses, and surveillance typically focuses on laboratory-confirmed cases rather than those diagnosed based on clinical suspicion.
The CDC warns that untreated or underdiagnosed Lyme disease can result in severe, long-term complications, including neurological, cardiac, and joint issues.
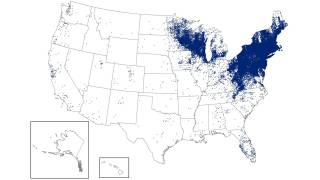
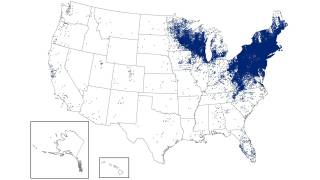
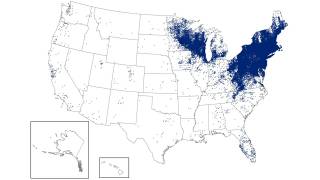
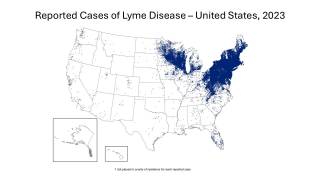
In the U.S., over 89,000 cases of Lyme disease were reported in 2023, but it is estimated that 476,000 people were actually infected.
Globally, infections are increasing in Europe, Asia, and Australia, as warmer temperatures allow ticks to thrive in new areas and remain active for longer periods.
An innovative company recently addressed this need by detecting antibodies to nine Lyme-associated Borrelia species, far more than the one or two species covered by most conventional tests, including the CDC's two-tier protocol (ELISA followed by Western blot).
AcuDart Health maintains worldwide access to its at-home Tick-Borne Disease (TBD) Screening Test Panel. The company stated in a press release that AcuDart's panel provides a convenient, accurate option for early screening.
Initially launched in the United States in August 2024 and now available in 51 countries, AcuDart Health's Tick-Borne Disease Test Panel screens for Lyme disease, Tick-Borne Relapsing Fever, Bartonellosis, and Babesiosis without requiring a doctor's authorization.
In a significant update announced on February 24, 2026, the company's Lyme disease tests received FDA clearance for the IgG kit in August 2024 and the IgM kit in June 2025, enhancing the credibility of its at-home diagnostics.
The test utilizes a simple finger-prick blood sample collected at home, shipped in lightweight, environmentally friendly pouches, and processed by partner laboratory IGeneX. Results are delivered digitally in 3-5 business days.
A few drops of blood are needed to test for nine species of Borrelia, the bacteria that cause Lyme disease. The sample collection card is an alternative to an SST tube. Red blood cells are separated without a centrifuge, enabling the laboratory to perform tests on dry blood.
The panel evaluates antibodies to 19 markers across the four diseases, providing broader coverage than conventional lab tests.
The company claims sensitivity above 83%, higher than PCR, ELISA/Western blot, and many commercial alternatives, potentially reducing false negatives. Other tests with a higher sensitivity than AcuDart are the ImmunoBlots from IGeneX.
However, the CDC says that a negative test does not rule out Lyme disease, particularly early Lyme disease, as antibodies can take a while to rise after infection begins. Furthermore, the company advises that positive results should be followed up with healthcare professionals for confirmation and treatment.
From a Lyme Disease prevention option, one vaccine candidate is progressing in phase 3 clinical trials in 2026, but is not commercailly available as of February 25, 2026
Our Trust Standards: Medical Advisory Committee