New TB Vaccine Candidates Show Limited Protection in Indian Clinical Trial
Two experimental tuberculosis (TB) vaccines, VPM1002 and Immuvac, were found to be safe and well-tolerated in a major Phase 3 trial involving thousands of healthy household contacts of smear-positive TB patients in India.
However, neither vaccine significantly reduced the overall incidence of microbiologically confirmed TB or pulmonary TB.
The PreVenTB trial results, published in The BMJ on April 9, 2026, showed that the efficacy of VPM1002 was 21.4% against all forms of TB and 19.5% against pulmonary TB, both of which were not statistically significant.
However, VPM1002 showed 50.4% efficacy (95% confidence interval of 0.8% to 75.2%) against extrapulmonary TB.
Furthermore, Immuvac demonstrated 33.2% efficacy against extrapulmonary TB, which was also not significant.
A post-hoc analysis revealed particularly strong results in children aged 6 to 14 years, with VPM1002 showing an efficacy of 64.6% against all forms of TB.
Both vaccines performed better against extrapulmonary TB in participants with positive tuberculin skin tests, showing around 65% efficacy, and were well tolerated, with mild local reactions occurring in about one-third of recipients. They also induced specific immune responses, such as polyfunctional CD4+ T cells. However, they did not prevent latent TB infection overall.
The study authors concluded that while the vaccines are safe, they do not protect against all forms of TB, including pulmonary TB. VPM1002 showed meaningful efficacy against the more challenging-to-diagnose extrapulmonary form, especially in certain subgroups.
For context, the existing BCG vaccine remains the only licensed TB vaccine worldwide.
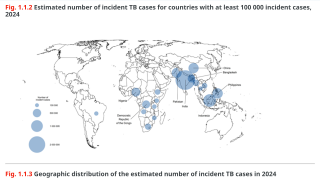
As of 2026, various BCG strains are routinely administered in approximately 156 of 194 countries with moderate-to-high TB incidence. Common strains include Danish 1331, Bulgarian SL-222, Russian, and Tokyo 172-1, and about 10 different substrains have been documented globally, with the Danish strain being the most widely used in many regions.
In 2023, approximately 323 million BCG doses were administered worldwide, primarily to newborns to protect against severe forms of childhood TB.
In the United States, BCG is not part of the routine immunization program due to low TB incidence. It is used only selectively for high-risk individuals, such as certain travelers or infants with ongoing exposure.
Recently, the BCG vaccine has been deployed in numerious cancer treatments.
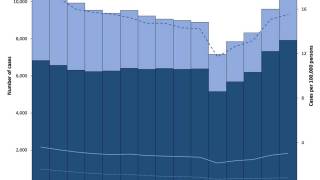
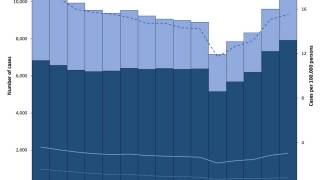
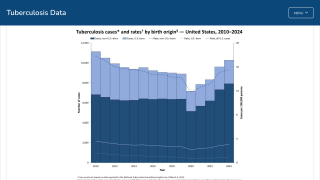
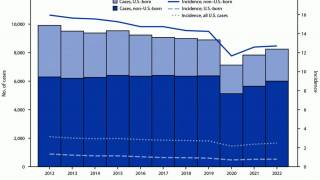
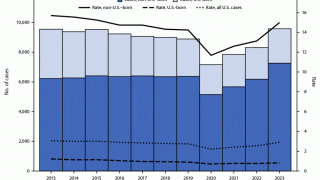
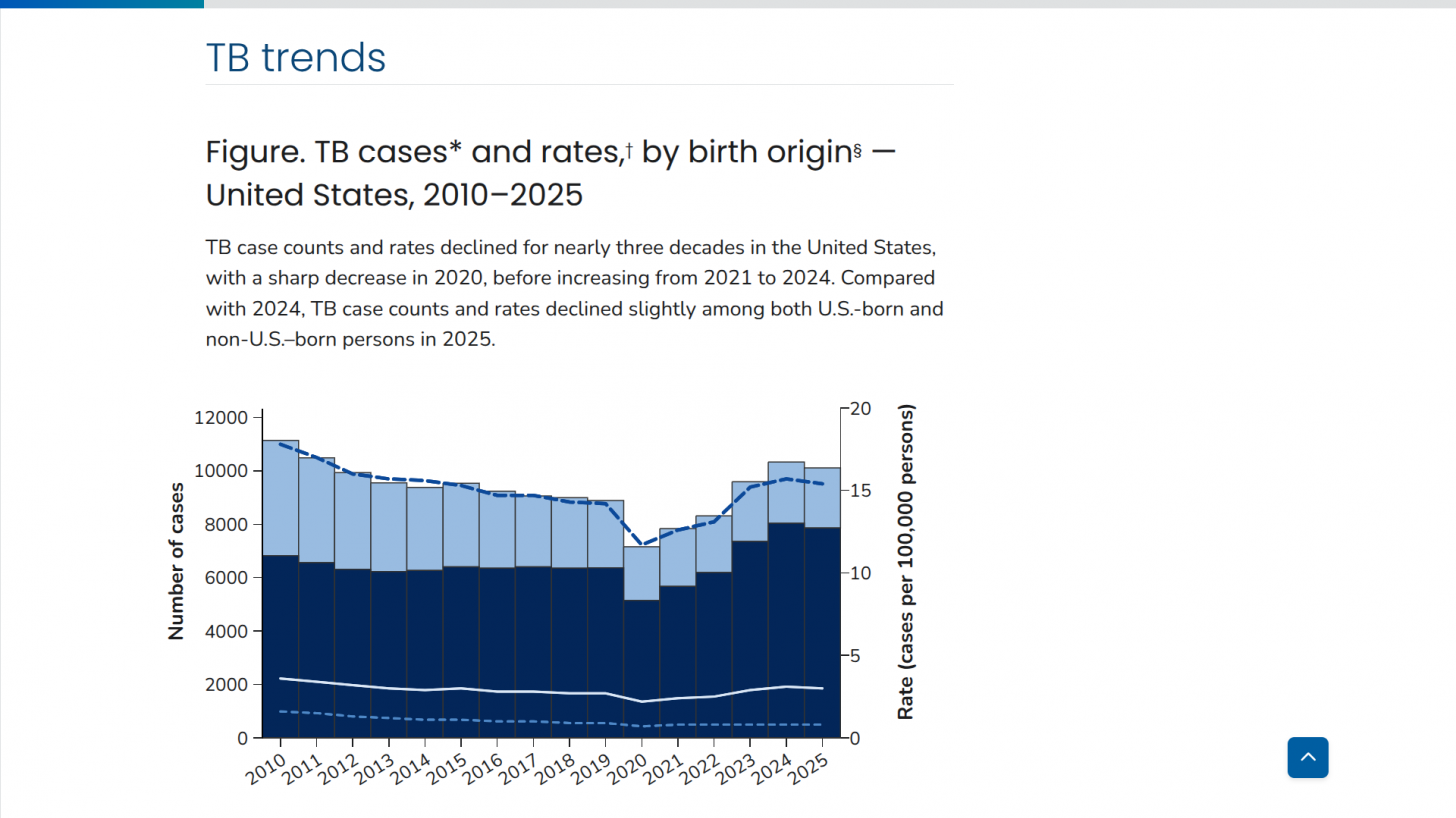
According to new data from the U.S. CDC, TB cases in the U.S. over the past five years show a sharp drop in 2020, followed by a steady rebound from 2021 through 2024, and a small decline in 2025. Despite the recent uptick, the overall incidence rate remains among the lowest in the world.
This vaccine clinical trial was funded by the Indian Council of Medical Research and registered as CTRI/2019/01/017026.
Our Trust Standards: Medical Advisory Committee