Near-Point-of-Care Molecular Tests for Diagnosing Tuberculosis Recommended
The World Health Organization (WHO) has issued its first recommendations for new near-point-of-care (NPOC) molecular tests for diagnosing tuberculosis (TB).
These WHO recommendations include using easy-to-collect tongue swab samples to simplify and expand access to testing, as well as a cost-saving sputum pooling strategy to enhance testing efficiency for both TB and rifampicin-resistant TB.
"These new WHO recommendations represent a significant advancement in making TB testing faster and more accessible," said Dr. Tereza Kasaeva, Director of WHO's Department for HIV, TB, Hepatitis, and STIs, in a press release issued on March 9, 2026.
"The WHO urges countries and partners to collaborate in implementing these guidelines to address ongoing diagnostic gaps and ensure that everyone with TB is diagnosed early and can start life-saving treatment without delay."
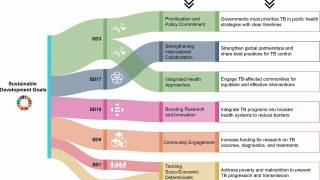
Under the WHO's End TB Strategy and the political declaration from the United Nations High-Level Meeting on TB, countries committed to providing early diagnosis of TB and universal access to WHO-recommended rapid molecular tests.
However, critical diagnostic gaps remain, as millions of people still experience delays in diagnosis or miss it entirely due to systemic barriers.
These barriers include a continued reliance on sputum as the sample type, which not everyone with TB can produce; the limited availability of laboratory-based tests that are not always accessible where patients seek care; and the high costs of tests and associated equipment, which hinder the expansion of testing networks.
To assist countries in strengthening TB disease and drug resistance detection, WHO provides evidence-based policy guidance on TB testing, which is routinely updated.
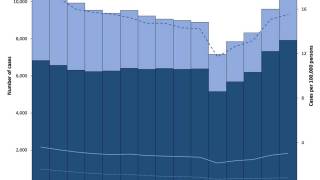
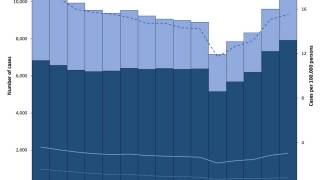
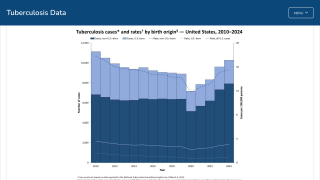
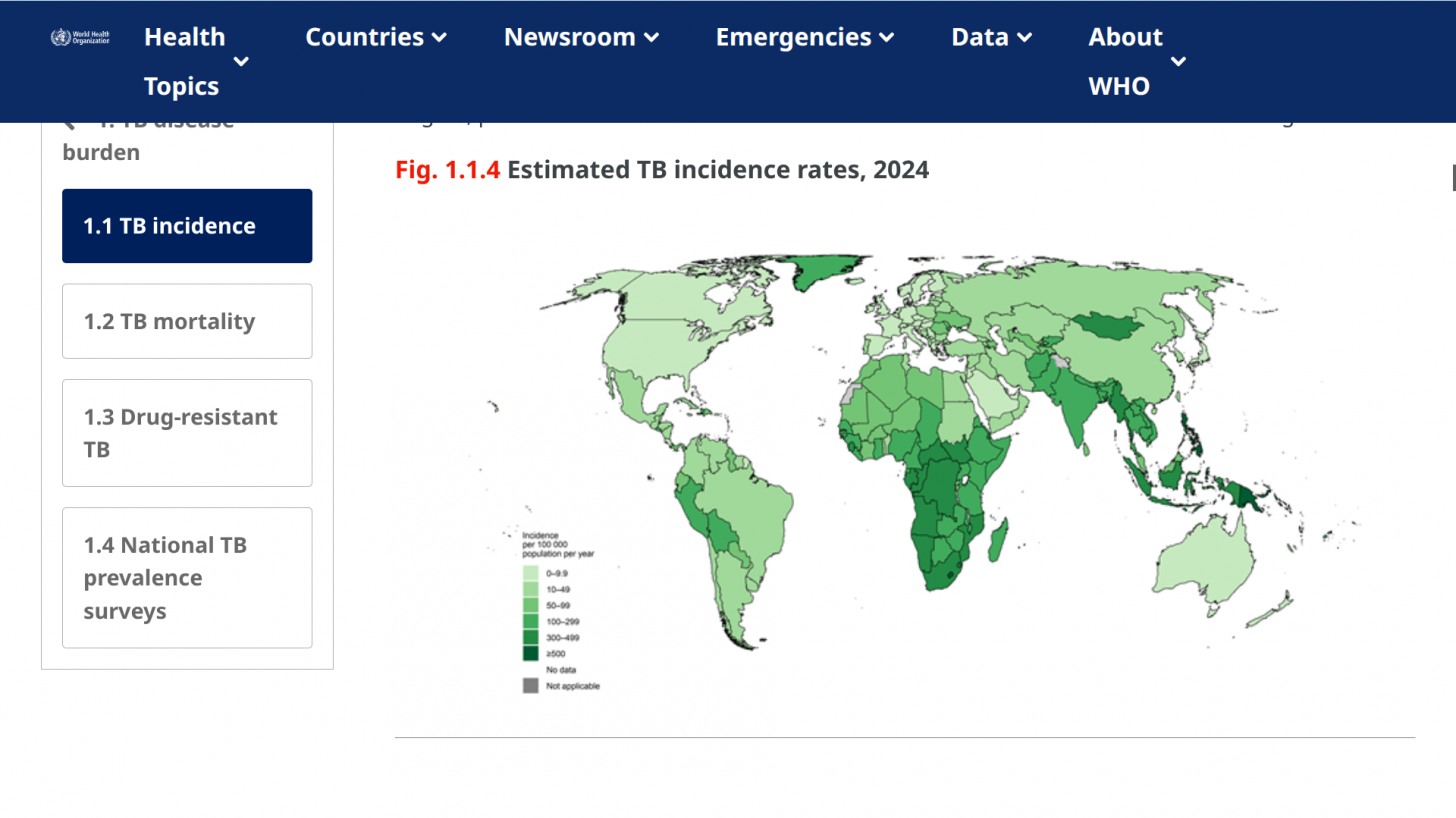
According to WHO data, eight countries, led by India, account for about 67% of the global total of new TB cases in 2024.
The complete policy for the diagnosis of TB and drug-resistant TB will be released this year in the WHO consolidated guidelines on tuberculosis. Module 3: Diagnosis. 2nd edition. The summary of findings and the evidence-to-decision tables will be produced in accordance with the GRADE method and made available on the WHO website.
As of March 10, 2026, the WHO's primary and longstanding recommendation for TB vaccination remains the 100-year-old Bacille Calmette-Guérin (BCG) vaccine, which has over ten versions used by various countries.
BCG vaccine candidates such as M72/AS01E, VPM1002, and MTBVAC are currently in late-stage clinical trials.
Our Trust Standards: Medical Advisory Committee