Spain and the UK Were Wrong; Gonorrhea Remains Not Vaccine-Preventable

Recent findings from the world's largest randomized controlled trial (RCT) have confirmed that the meningococcal B vaccine 4CMenB (Bexsero®) does not protect against gonorrhea, contradicting earlier optimism based on observational studies.
This news, published on February 25, 2026, has led experts to conclude that the disease is not preventable by currently available vaccines.
The findings were presented last week at the Conference on Retroviruses and Opportunistic Infections by researchers from Griffith University's Institute for Biomedicine and Glycomics and the Kirby Institute at UNSW Sydney.
The double-blind, placebo-controlled GoGoVax trial enrolled 587 individuals with a recent history of gonorrhea or syphilis. Over the two-year study period, the incidence of gonorrhea was nearly the same in both study groups: 48.1 cases per 100 person-years in the vaccine group compared to 47.8 in the placebo group (incidence rate ratio 1.01, 95% CI 0.80–1.26).
This resulted in a vaccine efficacy of –0.5% (95% CI –26% to +20%).
Lead investigator Prof. Kate Seib remarked in a media statement, "These results were unexpected given the observational findings and somewhat disappointing considering the need for a gonorrhea vaccine."
"Our data clearly shows that 4CMenB was not effective in reducing gonorrhea incidence among our participants."
This RCT's outcome aligns with the smaller French DOXYVAC RCT, which reported a non-significant 22% relative reduction in gonorrhea incidence (adjusted HR 0.78, p=0.061) using an open-label design.
In summary, earlier observational data, including meta-analyses that suggested an effectiveness of about 33–40% based on studies from New Zealand, the U.S., South Australia, and other locations, had generated significant enthusiasm and justified the UK's rollout of the vaccine for high-risk men in 2025.
However, observational studies are often unreliable for assessing vaccine effectiveness against sexually transmitted infections (STIs) due to factors like behavior, testing frequency, and partner networks that can obscure results. The two recent randomized trials—one showing borderline results and the other a clear negative outcome—have now dispelled that initial hope.
The UK program, which launched in August 2025 through sexual health clinics for individuals with recent STIs or multiple partners, will likely be reassessed, wrote these authors.
Galicia, Spain, was actually the first jurisdiction to implement a program to prevent gonorrhoea through vaccination.
Meanwhile, the U.S. Centers for Disease Control and Prevention (CDC) has never recommended 4CMenB for gonorrhea prevention, only for groups at risk for meningococcal disease.
The 4CMenB vaccine has been approved in about 40 countries and has had a significant impact on global health by reducing deaths and disabilities caused by N. meningitidis.
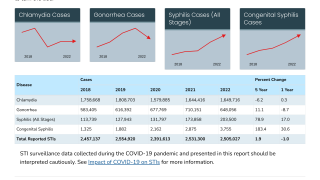
With over 82 million global gonorrhea cases annually, rising incidence, and antimicrobial resistance complicating treatment, CDC experts emphasize proven prevention: consistent condom use, regular STI testing/treatment, partner notification, and doxycycline post-exposure prophylaxis for select high-risk groups.
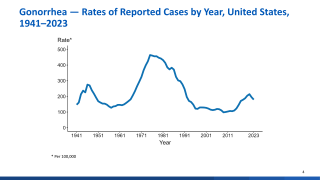
A dedicated gonorrhea vaccine is a critical unmet need, but until one arrives, gonorrhea is not vaccine-preventable as of March 2, 2026.
Our Trust Standards: Medical Advisory Committee