Topical Spray to Prevent and Treat New World Screwworm in Multiple Species Authorized

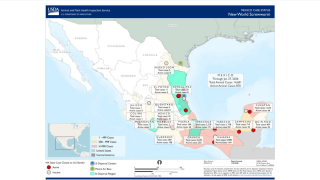
The New World screwworm (NWS) situation in Mexico shows ongoing activity in March 2026, but recent detections have been primarily in central states rather than near the Texas border.
Earlier in 2026, NWS cases were reported in Tamaulipas and Nuevo León.
To help address this threat to animals, the U.S. Food and Drug Administration (FDA) has recently issued an Emergency Use Authorization (EUA) for F10 Antiseptic Wound Spray with Insecticide, a topical solution containing benzalkonium chloride, polyhexanide, and cypermethrin.
This product is available over the counter without a prescription and is intended for the prevention and treatment of New World screwworm (NWS) infestations in animals.
To date, the FDA has conditionally approved or issued EUAs for other drugs to prevent and/or treat NWS in cattle, dogs, and cats. However, F10 Antiseptic Wound Spray with Insecticide is the first product authorized for NWS indications in additional species.
Most animals in the United States are at low risk for NWS due to their geographic location. However, animals near the U.S.-Mexico border or those that have traveled to countries with active NWS cases are at a higher risk of exposure.
As of March 10, 2026, the FDA has concluded that, based on available scientific evidence, it is reasonable to believe that F10 Antiseptic Wound Spray with Insecticide may be effective in preventing and treating NWS myiasis in a variety of animals.
These include cattle, horses, minor species of hoofed animals (such as sheep, goats, and deer), raptors and other wild birds, pet birds, and captive wild, exotic, and zoo mammals.
It is important to note that the known and potential benefits of using this product outweigh its known and potential risks.
Detailed product information about F10 Antiseptic Wound Spray with Insecticide can be found in the Fact Sheet: Emergency Use Authorization of F10 Antiseptic Wound Spray with Insecticide (benzalkonium chloride, polyhexanide, and cypermethrin topical solution) for NWS.
This EUA will remain in effect until it is revoked or the HHS Secretary terminates the declaration that the potential public health emergency presented by NWS justifies the emergency use authorization of animal drugs for NWS. F10 Antiseptic Wound Spray with Insecticide is sponsored by Health and Hygiene (Pty) Ltd, based in South Africa.
As of March 11, 2026, no NWS cases (human or animal) have been reported in the USA.
Furthermore, there are no specific FDA-approved drugs for treating NWS in humans.
The FDA says the primary treatment approach involves physically removing all larvae and eggs, which may necessitate surgical extraction if they are deeply embedded. This procedure is often combined with wound debridement, irrigation, and monitoring for secondary bacterial infections, which can be treated with antibiotics if necessary.
Currently, no vaccines are available for humans against NWS.
Our Trust Standards: Medical Advisory Committee