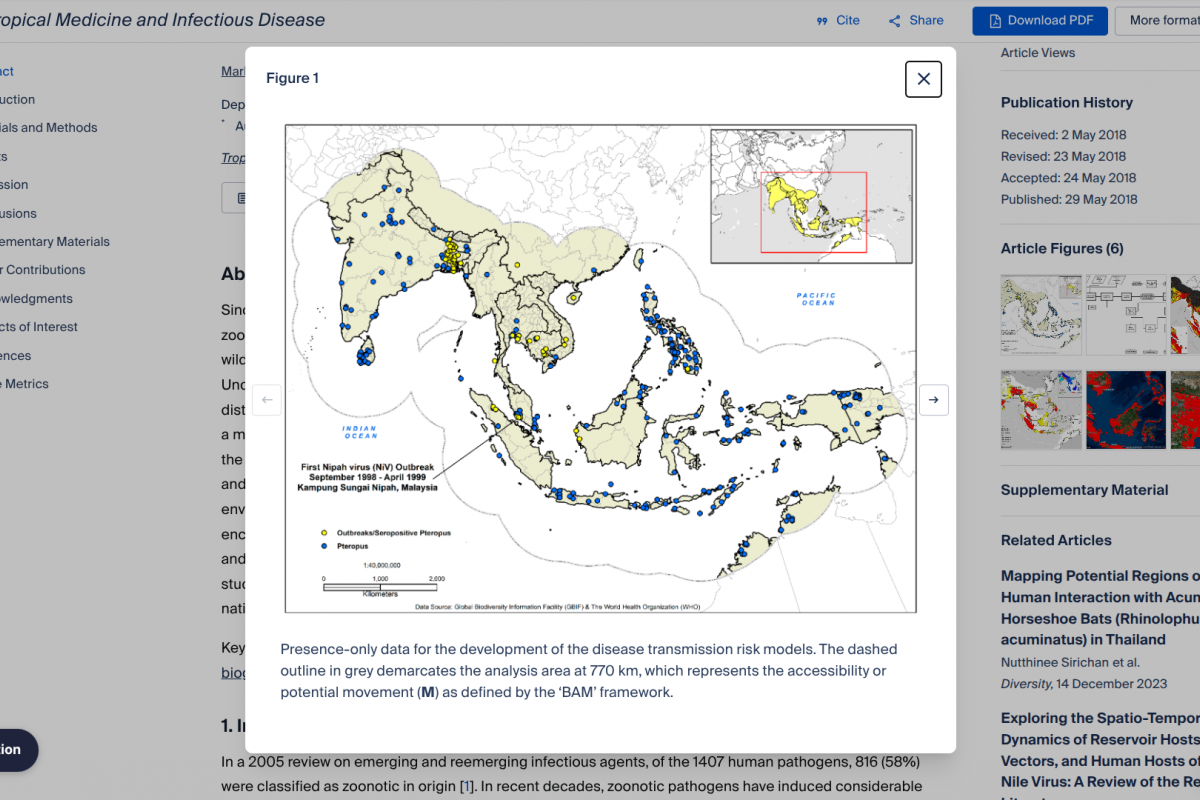
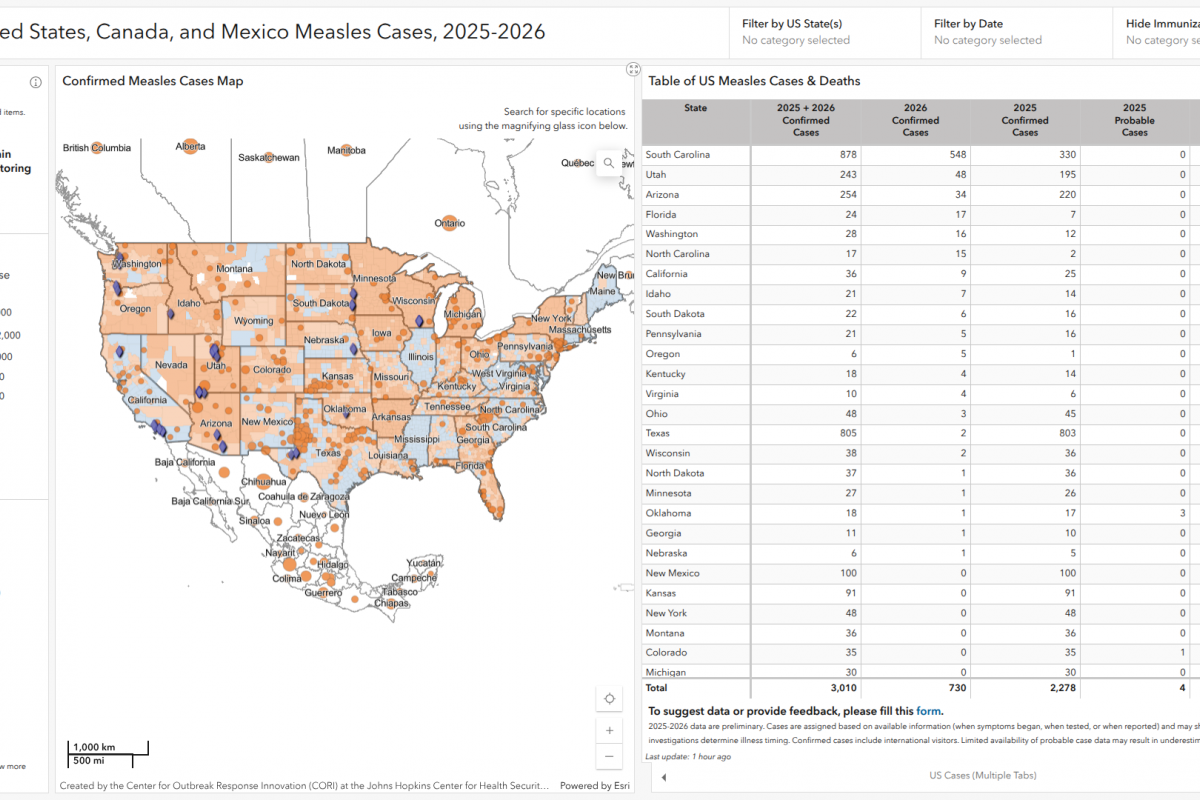
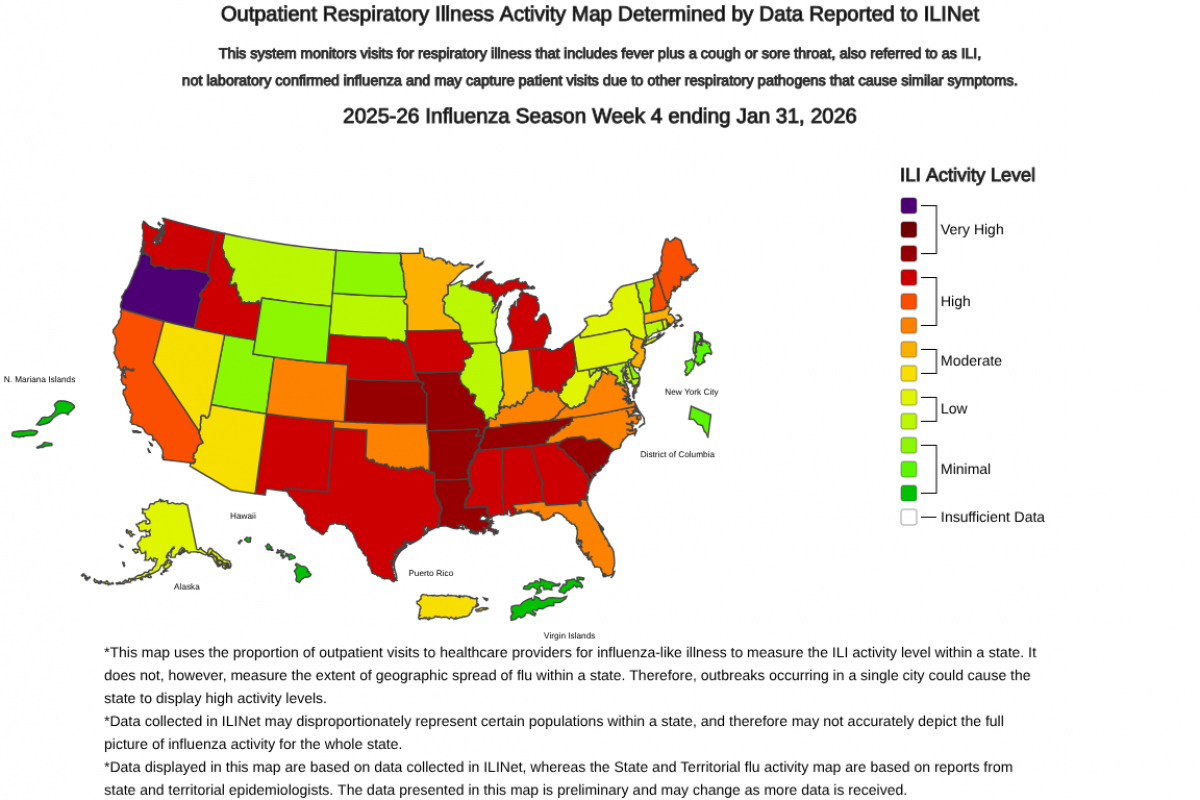
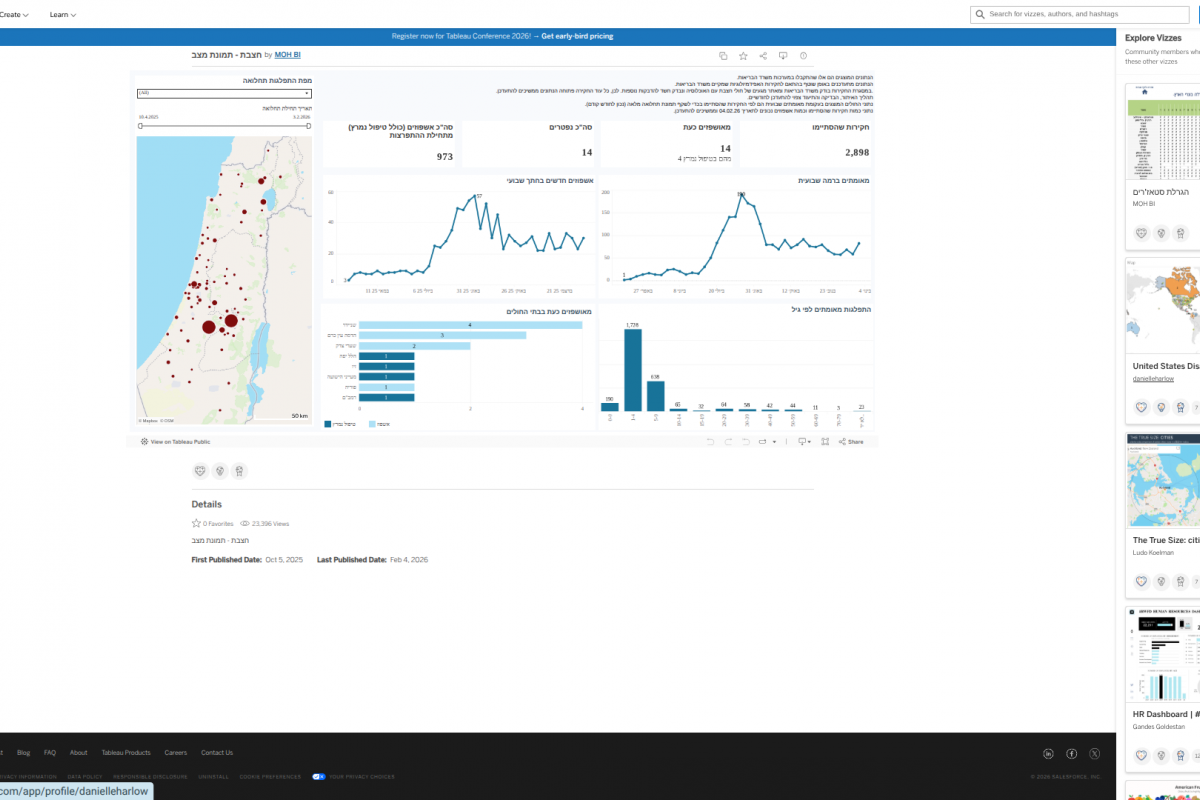
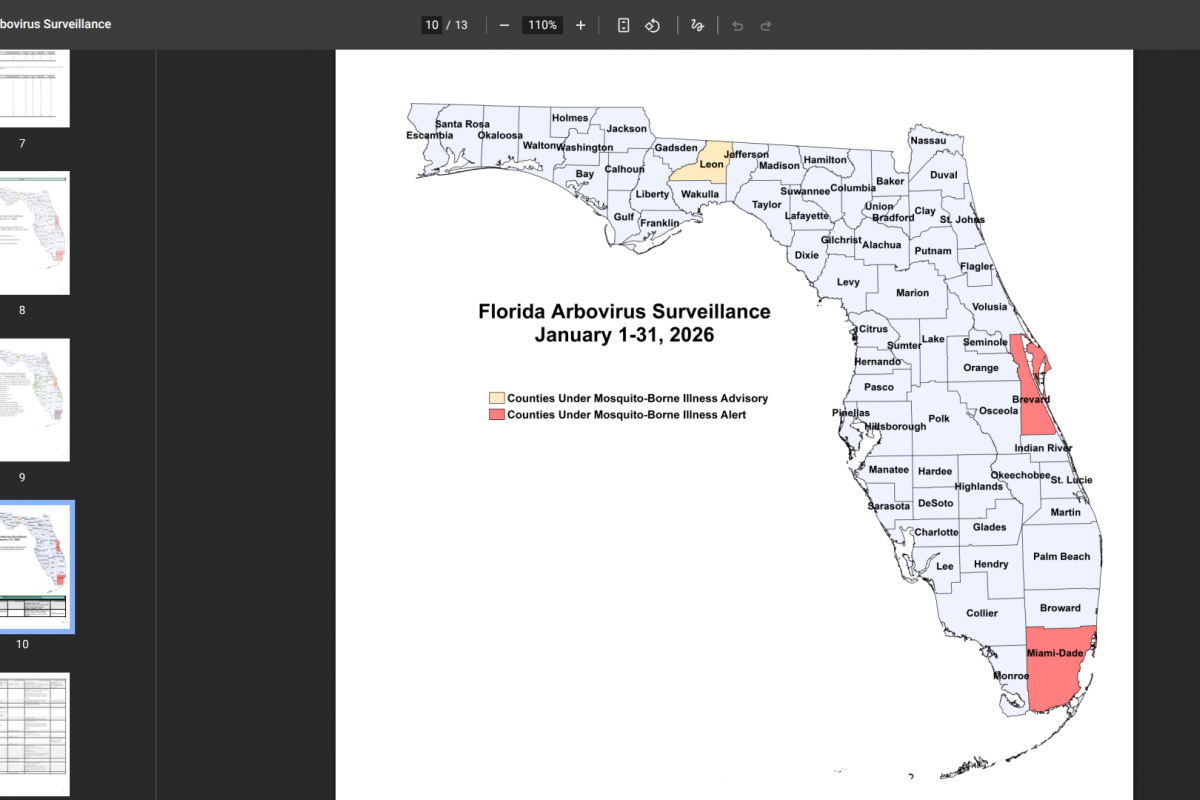
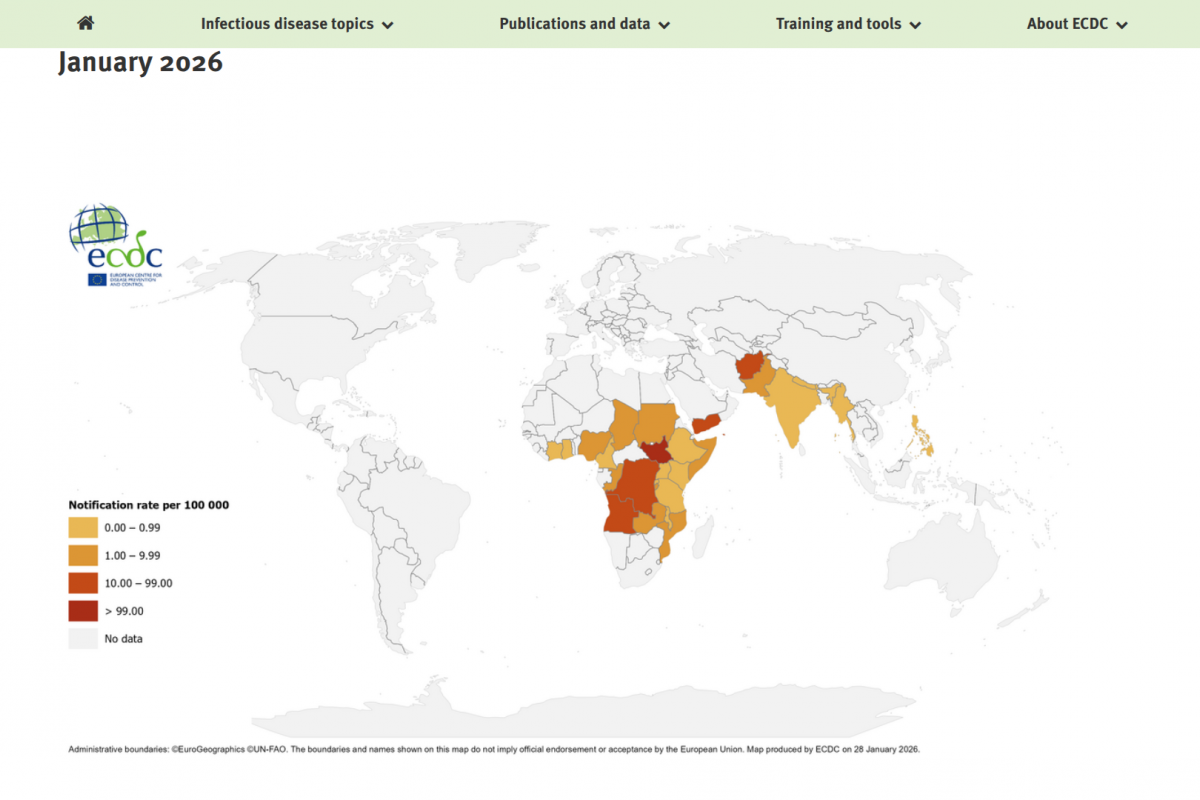
In the past year, the World Health Organization (WHO) reported over 600,000 cases of cholera or acute watery diarrhea and nearly 7,600 deaths across 33 countries.
However, these figures are likely underreported, as cholera cases often go unrecorded.
Today, Gavi, the Vaccine Alliance, UNICEF, and the WHO announced that the global supply of oral cholera vaccine (OCV) has increased sufficiently to resume life-saving preventive vaccination campaigns for the first time in over three years.
A first allocation of 20 million doses is being deployed for preventive campaigns.
Of these, 3.6 million doses were delivered to Mozambique; 6.1 million to the Democratic Republic of the Congo, which is also experiencing significant outbreaks; and 10.3 million doses are planned for delivery to Bangladesh.
"Global vaccine shortages forced us into a cycle of reacting to cholera outbreaks instead of preventing them. We are now in a stronger position to break that cycle. I thank EUBiologics, currently the only manufacturer producing cholera vaccines at the scale needed for mass vaccination campaigns, for its efforts, and urge others to enter this vital space. These vaccines will save lives," said Dr Tedros Adhanom Ghebreyesus, WHO Director-General, in a press release on February 4, 2026.
Over the last few years, the annual global supply of OCV has doubled from 35 million doses in 2022 to nearly 70 million doses in 2025. The doses are being financed by Gavi and procured and delivered to countries by UNICEF.
"The multi-year surge in cholera cases and resulting unprecedented demand for vaccines were stark reminders that sustainable, accessible vaccine supply is a global public good – and the world cannot afford complacency," added Dr Sania Nishtar, CEO of Gavi, the Vaccine Alliance.
"For the first time in years, this increase in vaccines will allow us to prevent large-scale cholera emergencies better," said Catherine Russell, UNICEF Executive Director. "Resuming preventive cholera vaccination will protect children and help stop this highly contagious disease in its tracks. But it must go hand in hand with other efforts, including better access to safe water and basic sanitation."
While global vaccine supply steadily improves, the one-dose strategy will remain the standard for outbreak responses, with the use of two doses considered on a case-by-case basis.
Cholera spreads through contaminated food and water, causing severe diarrhoea and dehydration. It can lead to death if it is not treated quickly. It is found in places without safe water and sanitation, mainly in localities affected by conflict and poverty.
The WHO says vaccination is only one aspect of cholera prevention and response. Long-term investments in safe water, sanitation, and hygiene infrastructure, alongside disease surveillance, rapid treatment, and community engagement, remain essential to prevent outbreaks from starting and spreading, and to reduce deaths in the long term.
In the United States, OCVs are offered at travel vaccine clinics, located in every state.