Cholera Vaccines
Cholera Vaccines February 2026
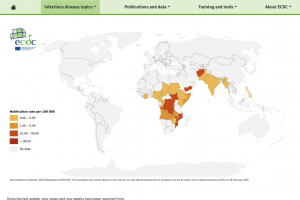
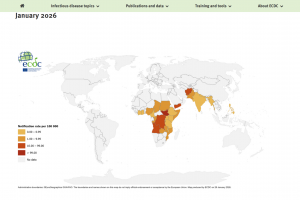
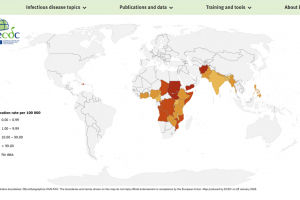
The U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), the World Health Organization (WHO), and the U.K. National Health Service (NHS) recommend oral cholera vaccines (OCVs) for travelers visiting countries experiencing outbreaks. As of February 2026, the WHO has prequalified Vaxchora®, Dukoral®, Shanchol™, Euvichol®, and Euvichol-S OCVs.
On February 4, 2026, the WHO stated Following sustained efforts by global agencies, manufacturers and partners, the annual global supply of OCV has doubled from 35 million doses in 2022 to nearly 70 million doses in 2025. A first allocation of 20 million doses in 2026 is being deployed for preventive campaigns. Of these, 3.6 million doses were delivered to Mozambique; 6.1 million to the Democratic Republic of the Congo, which is also experiencing significant outbreaks; and 10.3 million doses are planned for delivery to Bangladesh.
The WHO says that all OCVs require two vaccine doses for complete protection for up to three years, while a single dose provides short-term protection. GAVI reported in 2025 that, during the current cholera outbreak, only one OCV dose course had been implemented in reactive vaccination campaigns.
Dukoral® is administered with a buffer solution that requires 150 ml of clean water for adults. It can be administered to all individuals aged two and above.
Vaxchora® (lyophilized CVD 103-HgR) is a single-dose, oral vaccine approved by the U.S. FDA in June 2016. The safety and effectiveness of VAXCHORA have not been established in individuals with immunocompromised conditions. In August 2023, the U.S. Centers for Disease Control and Prevention (CDC) published "Cholera Vaccine: Recommendations," highlighting CVD 103-HgR Vaxchora for travelers going to areas of active toxigenic Vibrio cholerae O1 transmission.
Shanchol™ and Euvichol® are essentially the same vaccines produced by two different manufacturers. Euvichol-Plus® is a simplified formulation of the existing inactivated OCV jointly developed by Eubiologics and the International Vaccine Institute for cholera prevention. Euvichol®-S improves productivity by approximately 40% over Euvichol-Plus®. In January 2025, a batch of 948,500 doses of Euvichol-S arrived in Angola.
HILLCHOL® (BBV131) is a novel single-strain OCV developed by Bharat Biotech International Limited under HilleLaboratories'ries license and funded by Merck and Wellcome Trust. It is a two-dose vaccine that BBIL states needs to be administered orally on Day 0 and Day 14. HILLCHOL is suitable for individuals over one year old and is available in mono- or multidose formats.
Cholera Vaccine Supply 2026
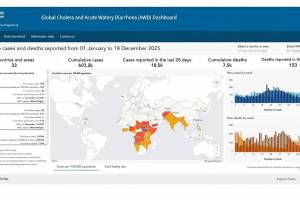
The International Coordinating Group (ICG) on Vaccine Provision has managed and coordinated the provision of OCV supplies and vaccines since 2013. Since the ICG's establishment in 1997, the WHO, UNICEF, and Médecins sans Frontières have facilitated the distribution of over 73 million doses of OCV to 23 countries. The OCV vaccine dashboard was updated for 2026.