Promising Antiviral Activity Against Dengue Announced

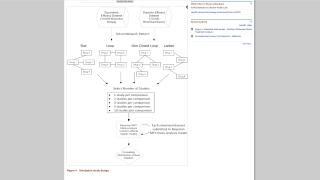
The Janssen Pharmaceutical Companies today confirmed at the American Society of Tropical Medicine & Hygiene Annual Meeting positive results from a Phase 2a human challenge study evaluating JNJ-1802, a first-in-class oral antiviral in development for the prevention of dengue.
Announced on October 20, 2023, the data showed that the JNJ-1802 induced antiviral activity against dengue (DENV-3) in humans, compared to placebo, and is safe and well-tolerated.
JNJ-1802 is the first antiviral to show such activity in humans during a clinical trial.
The compound is not a vaccine and has advanced to a community-based field study to establish efficacy against circulating dengue serotypes in a real-world setting. The new research is being conducted in 30+ sites in 10 countries.
Marnix Van Loock, Ph.D., Lead for Emerging Pathogens, Global Public Health R&D at Janssen Pharmaceutica NV, stated in a press release, “Dengue requires global action, and we are proud to collaborate alongside partners around the world in advancing the development of this compound to its next phase.”
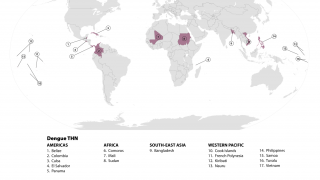
According to the U.S. CDC, dengue viruses are spread to people through the bite of an infected Aedes species. Annually, up to 400 million people get infected with dengue, and 40,000 die from severe dengue.
This new study follows data published in the journal Nature in March 2023, which showed that JNJ-1802 provides strong protection against dengue in non-human primates and mice.
And a Phase 1 first-in-human clinical study showed that the antiviral was safe and well-tolerated.
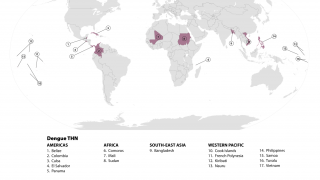
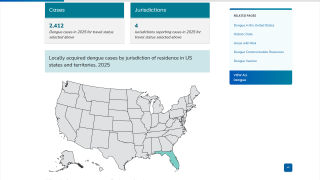
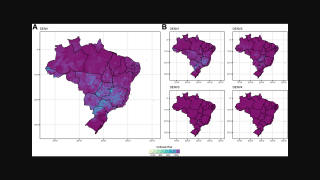
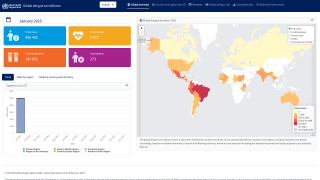
Globally, dengue outbreaks are active in numerous countries during 2023.
As of October 21, 2023, two dengue vaccines are approved for use in various countries.
Our Trust Standards: Medical Advisory Committee