Second-Generation Dengue Vaccine Withdraws From U.S.

The future vaccination plans for U.S. travelers and those living in dengue-outbreak areas, such as Puerto Rico, were disrupted yesterday.
Takeda announced on July 11, 2023, that the Company had voluntarily withdrawn the U.S. Biologics License Application (BLA) for its dengue vaccine candidate, TAK-003, following discussions with the U.S. Food and Drug Administration (FDA).
Takeda's press release stated that aspects of data collection could not be addressed within the current BLA review cycle.
On November 22, 2022, Takeda announced that the FDA had accepted and granted priority review of the TAK-003 BLA.
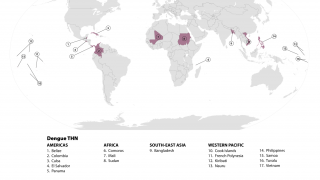
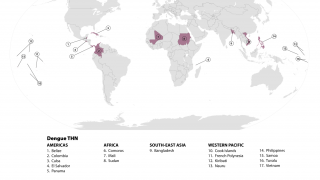
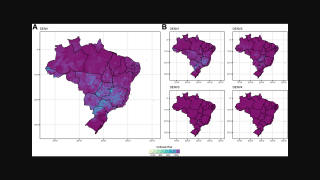
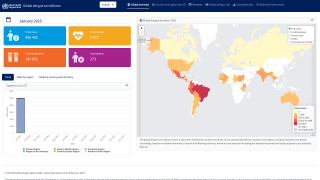
TAK-003, known internationally as QDENGA®, is approved in multiple endemic and non-endemic countries, such as the United Kingdom, Europe, and Brazil.
Gary Dubin, M.D., president of Takeda's Vaccines Business Unit, commented in a related press release, "The urgent global need to combat the growing burden of dengue remains, and we will continue to progress regulatory reviews and provide access for people living in and traveling to dengue-endemic areas while we work to determine next steps in the U.S."
QDENGA® is a tetravalent dengue vaccine preventing Dengue Fever or Severe Dengue caused by any of the four serotypes of the dengue virus.
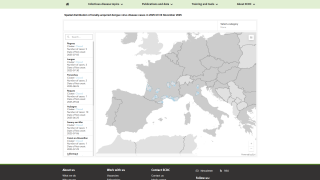
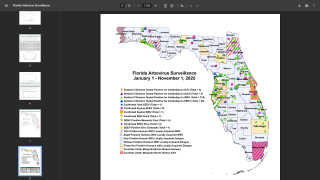
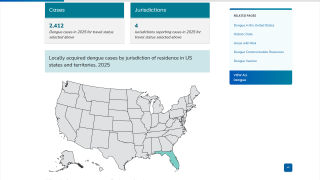
During 2023, locally-acquired dengue has been confirmed in Florida and Texas.
While other second-generation dengue vaccine candidates are in development, the initial FDA-approved dengue vaccine Dengvaxia® remains available in the U.S. but has specific pre-vaccination requirements.
Our Trust Standards: Medical Advisory Committee