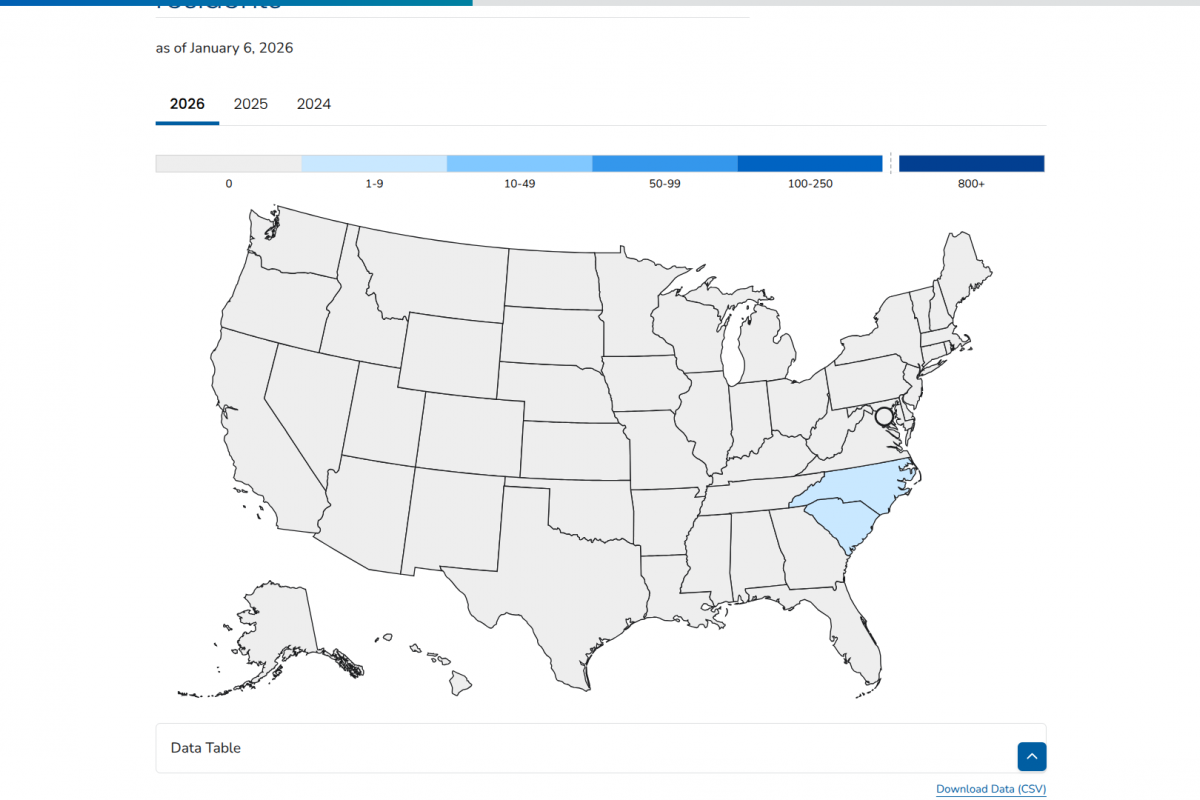
As of early 2026, cases of tick-transmitted Lyme disease continue to be reported in various European countries.
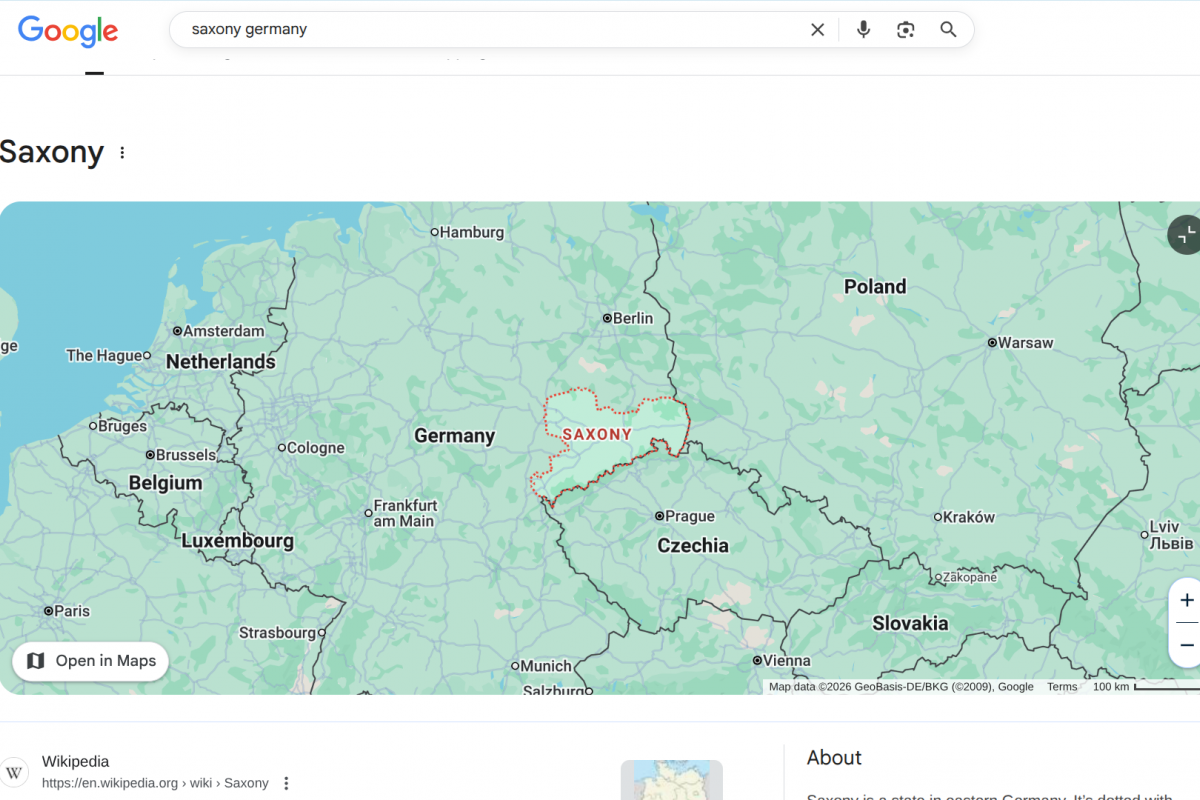
Recently, health authorities in the Free State of Saxony, Germany, noted a significant increase in Lyme disease cases in 2025.
According to the Saxon State Office for Health and Veterinary Affairs, a total of 2,623 infections were reported as of December 29, 2025.
This data marks a 61% increase from the 1,626 cases recorded in 2024, and continues an upward trend from 1,484 cases in 2023.
The data rise aligns with reports of elevated tick-borne infections across multiple German federal states.
During 2016−2020, nine federal states in Germany reported 63,940 LB cases, averaging 12,789 cases annually.
Fortunately, cases of tick-borne encephalitis, another serious tick-transmitted viral illness, decreased in Saxony in 2025 to 47 cases from 63 in 2024.
Currently, there is no approved vaccine for Lyme disease in humans in Europe and the United States.
However, promising developments are underway as Pfizer Inc. and Valneva SE are advancing their candidate VLA15, a multivalent protein subunit vaccine targeting the outer surface protein A of Borrelia bacteria.
VLA15 aims to block transmission from ticks to humans and covers prevalent serotypes in the USA and Europe.
Initially diagnosed in Lyme, Connecticut, ticks have spread this severe disease throughout the northeast sections of the USA.
As of January 12, 2026, these companies say 'regulatory filings are planned for 2026, subject to positive final clinical trial results.'