Lyme Disease Surge 2026: Europe and USA Brace for Increasing Cases
As Spring 2026 approaches in the Northern Hemisphere, health experts are preparing for another active Lyme disease season, following the upward trends in Europe and the United States observed in 2025.
According to the European Centre for Disease Prevention and Control (ECDC), Lyme disease, a Tickborne illness caused by the bacterium Borrelia burgdorferi, continues to expand its geographic presence in the United States and Europe.
This expansion is driven by factors such as climate change, urban sprawl, and increased outdoor activities.
While definitive case numbers for 2026 are still emerging, early projections suggest a potential increase over last year's figures, which already showed significant rises in key regions.
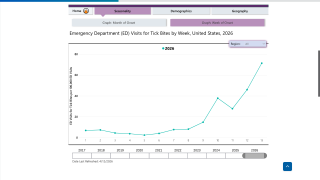
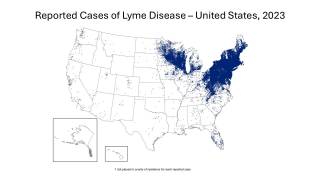
In the U.S., Lyme disease remains the most prevalent vector-borne illness, with cases peaking during the warmer months from May to August. The Centers for Disease Control and Prevention (CDC) reported over 89,000 confirmed cases in 2023; however, estimates based on insurance claims and other data suggest that the actual number of diagnosed and treated cases could be as high as 476,000 annually.
For 2025, while comprehensive national data is still being compiled, preliminary indicators point to continued growth. In 2025, the number is likely to approach or exceed 500,000, underscoring the growing burden of this disease, the CDC says.
Looking ahead to 2026, projections forecast further expansion due to warmer temperatures extending tick activity seasons. AI models integrating environmental and socioeconomic factors predict ongoing increases, with potential case counts surpassing 2025 levels by 5-10% in endemic areas.
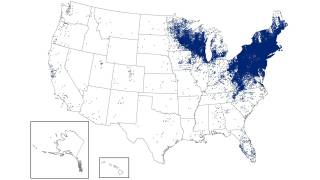
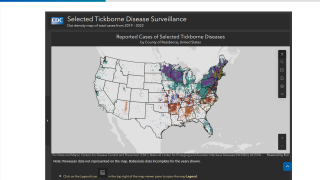
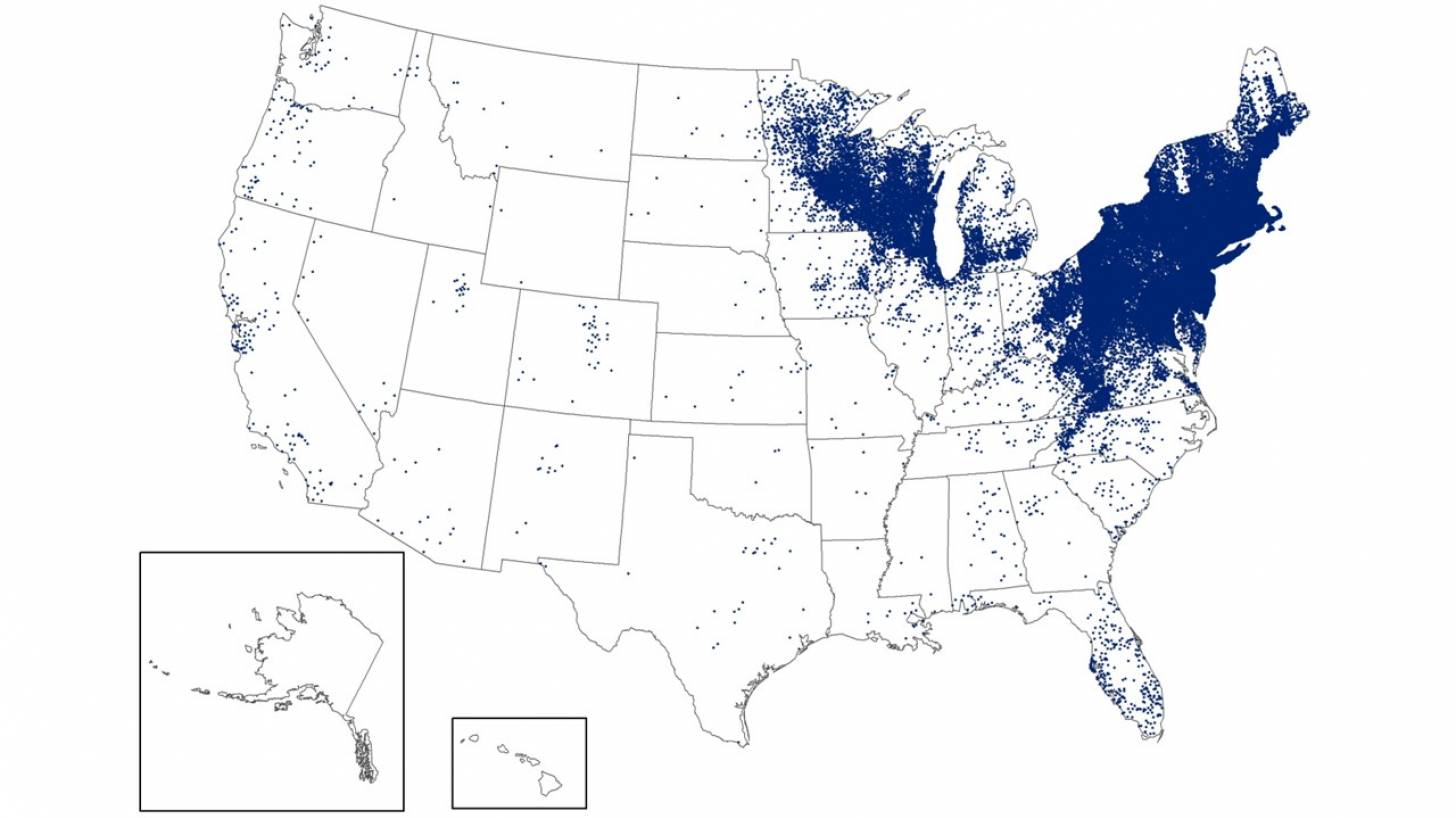
CDC experts attribute this increase to the expanding tick populations in the Northeast, Mid-Atlantic, and Upper Midwest, with emerging risks in southeastern states.
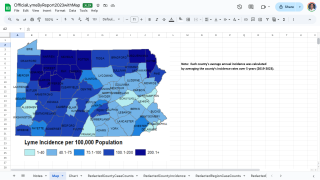
Across Europe, the ECDC estimates that Lyme disease (Lyme borreliosis) affects an estimated 132,000 people annually, based on surveillance data from 2015-2023; underreporting may raise the true figure.
High-incidence regions like Estonia, Finland, and Slovenia, impacting about 223 million residents—30% of Europe's population.
In 2025, several countries noted alarming spikes. Germany, a hotspot, saw significant rises: Saxony reported 2,623 cases by December 29, a 61% increase from 1,626 in 2024.
Bavaria tallied 4,156 cases through week 35, up from 3,126 in the same period of 2024.
Berlin confirmed 206 cases by week 24, signaling urban encroachment.
For 2026, ECDC experts anticipate sustained or heightened activity, with over 200,000 cases possible across the continent.
Meanwhile, amid rising case numbers, health agencies turn their attention to prevention. Currently, no approved human vaccine exists for Lyme disease, but Valneva SE's VLA15, a multivalent candidate, has advanced furthest in trials.
Targeting six serotypes of Borrelia prevalent in North America and Europe, VLA15 showed strong immunogenicity and a favorable safety profile in Phase 2 studies, including robust antibody responses after booster doses.
The pivotal Phase 3 VALOR trial completed primary vaccinations in late 2025, with participants monitored through the 2025 season. Phase 3 outcomes are expected in the first half of 2026.
Peter Bühler, Valneva's Chief Financial Officer, commented in a press release on February 19, 2026, "As we enter 2026, we look forward to the potentially transformative Phase 3 data readout for our Lyme disease vaccine candidate, while remaining focused on prudent capital allocation, to support shaping and growing our business in line with our strategic vision."
Subject to positive results, Valneva's partner Pfizer plans to submit a Biologics License Application to the U.S. FDA and a Marketing Authorization Application in Europe in 2026.
Regulatory reviews could follow shortly after, potentially leading to approvals by late 2026 or early 2027, marking a major step toward combating this endemic threat.
Our Trust Standards: Medical Advisory Committee