Health authorities in Venezuela are intensifying their efforts to combat a persistent yellow fever outbreak that began in 2025 and has continued into 2026.
As of late February 2026, Venezuelan health officials have confirmed 36 laboratory-verified human cases since the outbreak began, with at least six additional cases reported in the first seven weeks of 2026 alone, including one death.
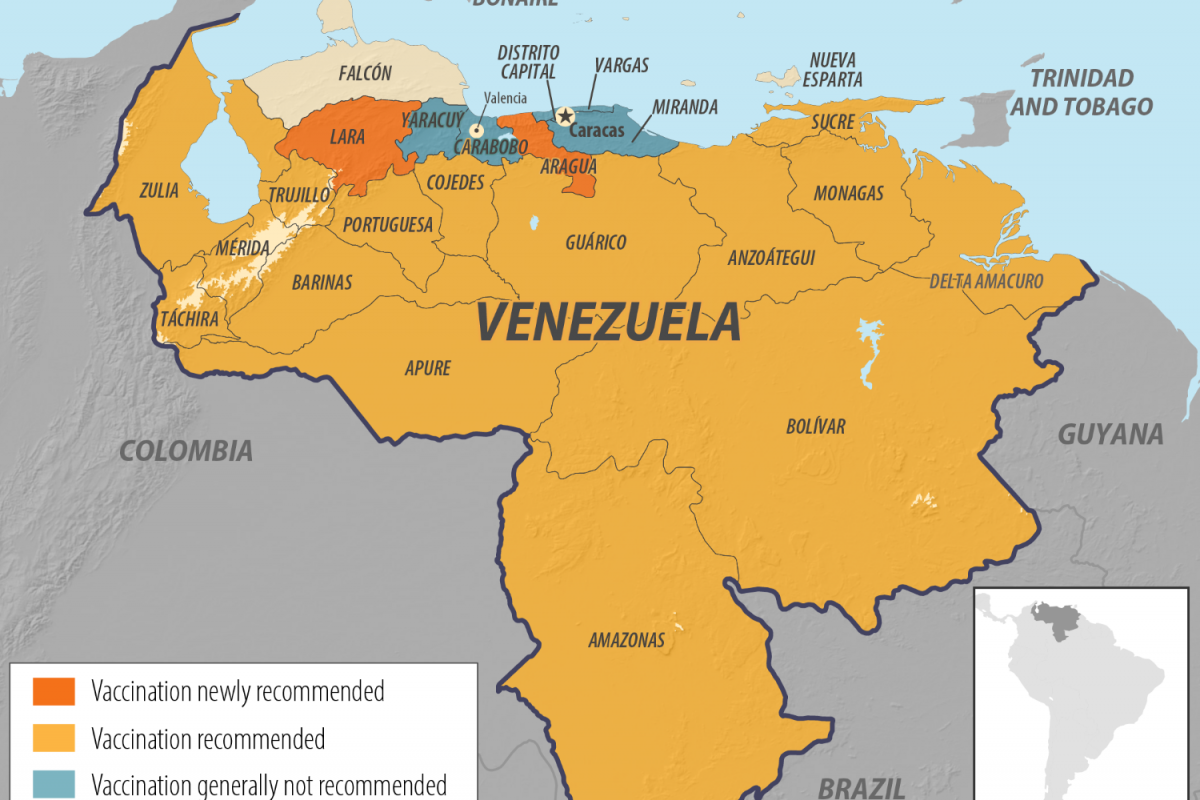
According to recent reports from the Pan American Health Organization (PAHO) and Venezuelan officials, the outbreak has affected 14 states, marking a significant geographic expansion compared to previous localized incidents.
These confirmed cases have been reported across a wide area of the country. New areas affected include states such as Aragua, Barinas, Lara, and Portuguesa, which were not traditionally considered high-risk zones. This change has raised concerns about shifting transmission patterns, potentially influenced by environmental factors, reduced vaccination coverage in some regions, and population movements.
Venezuela's outbreak occurs amid broader regional challenges in South America, where 346 confirmed cases and 143 deaths were reported in 2025 across seven countries, such as Bolivia, Colombia, and Peru.
The PAHO says recent regional reports indicate that the case fatality rate hovers around 40-44%, highlighting the virus's lethality among those who develop serious illness.
In response, Venezuela's Ministry of People's Power for Health has launched a nationwide emergency vaccination drive using Stamaril (YF-VAX). Health Minister Nuramy Gutiérrez emphasized that a single dose of the yellow fever vaccine provides lifelong immunity.
As of March 16, 2026, the U.S. Centers for Disease Control and Prevention (CDC) maintains a Level 2 - Practice Enhanced Precautions travel notice for yellow fever in Venezuela. They advise travelers to get vaccinated at least 10 days before visiting at-risk areas, especially those located below approximately 2,300 meters (7,550 feet) in elevation.
The CDC advises travelers planning to visit Venezuela or other outbreak areas in 2026 to consult a healthcare provider or travel vaccine clinic well in advance, as not all facilities stock the yellow fever vaccine.