For Next RSV Season, Younger People Have Access to Approved Vaccine
In preparation for the next Respiratory Syncytial Virus (RSV) season, which generally begins in Florida, the U.S. Food and Drug Administration (FDA) recently expanded the age range for Arexvy, an approved vaccine.
This GSK vaccine now includes adults aged 18 to 49 who are at increased risk for lower respiratory tract disease (LRTD) from RSV.
Previously, this vaccine was approved for most adults aged 60 and older, as well as those aged 50 to 59 with underlying health conditions who face heightened risks. It's important to note that this vaccine isn't designed for use by pregnant individuals.
Sanjay Gurunathan, GSK's Head of Vaccines and Infectious Diseases Research and Development, expressed optimism about the expansion in a press release issued on March 13, 2026, saying, "This age extension addresses a significant need for adults in the U.S. who are at higher risk for severe RSV disease due to certain medical conditions."
"It also helps alleviate pressure on our healthcare system."
GSK stated it is advancing its regulatory submissions globally to expand the availability of its RSV vaccine and support long-term growth goals.
According to the U.S. CDC, RSV can have a notable impact on younger adults as well, with the annual estimates showing around 17,000 hospitalizations, 277,000 visits to emergency departments, and a staggering 1.97 million outpatient visits among those aged 18 to 49.
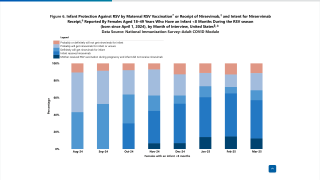
As of February 22, 2026, the CDC reported that 43.2% of adults aged 75 years and older and 32.8% of adults aged 50─74 years with a high-risk condition for RSV reported having ever received an RSV vaccine.
RSV vaccines are available throughout the USA at various clinics and pharmacies.
Our Trust Standards: Medical Advisory Committee