Are Flu Shot Boosters Needed in 2026
As the 2025-2026 influenza season comes to an end, the U.S. government recently released early data regarding the effectiveness of this year's flu shot.
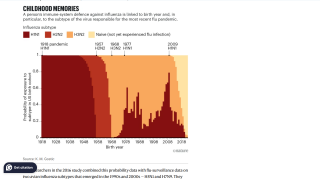
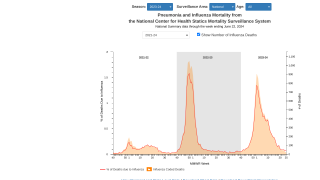
On March 12, 2026, the U.S. CDC's Morbidity and Mortality Weekly Report (MMWR, 75(9);124–128) noted that influenza vaccine effectiveness (VE) can vary each year based on several factors, including the antigenic similarity between the vaccine and circulating viruses, as well as patient characteristics. VE can be estimated using data from paired laboratory surveillance and vaccination records.
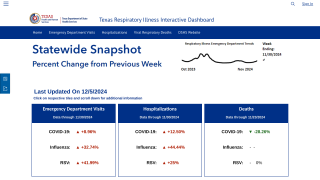
According to the MMWR, the estimated VE against laboratory-confirmed influenza for all age groups in California during the period from October 2025 to January 2026 was 33% (32% effectiveness against influenza A and 47% against influenza B).
The CDC stated that these results suggest that influenza vaccination is associated with a decreased likelihood of laboratory-confirmed influenza among both children and adults.
Furthermore, the CDC recommends annual influenza vaccination for all individuals aged 6 months and older to reduce the risk of influenza and its associated adverse health outcomes.
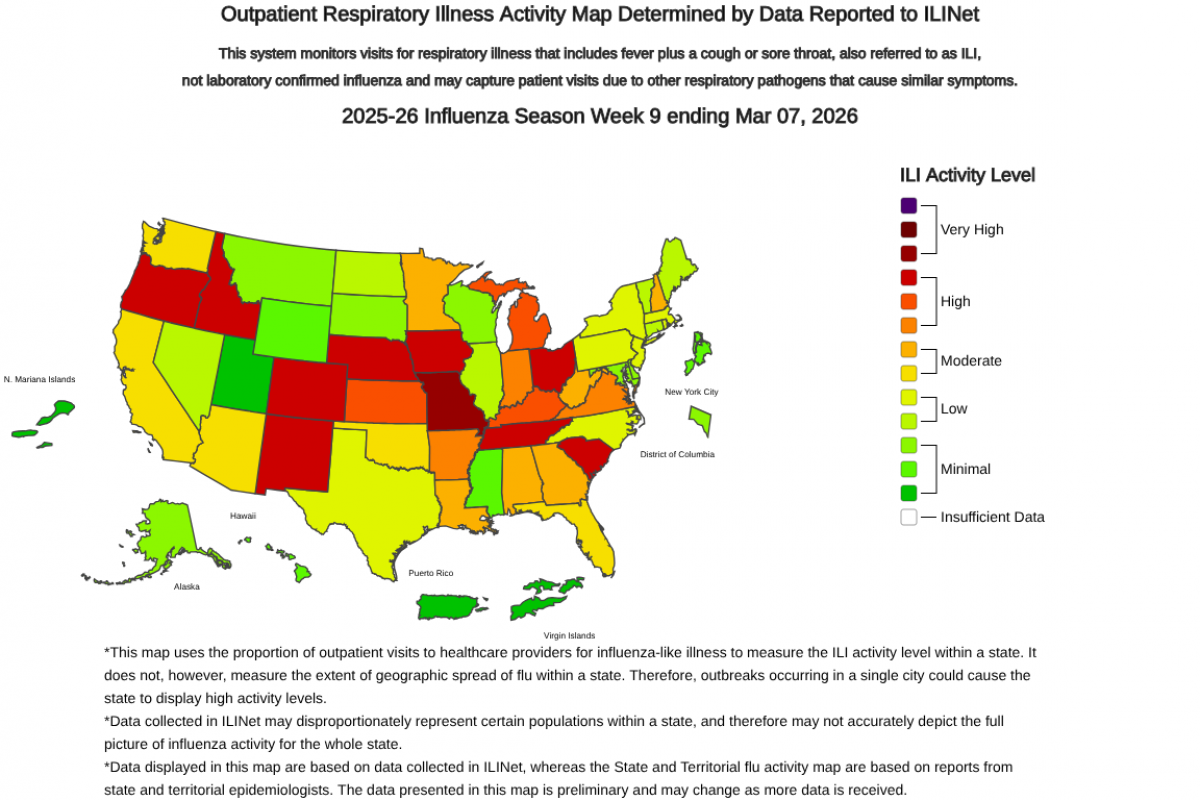
Additionally, some individuals located in high-risk areas may consider receiving a second flu shot in the later stages of flu season.
For the fast-approaching 2026-2027 flu season, the Vaccine and Related Biological Products Advisory Committee recently endorsed recommendations for viral strains to include in flu shots.
Our Trust Standards: Medical Advisory Committee