Lyme Disease Vaccine Has New Name, Continues Path to Approval
With Lyme disease season soon to spike in the Northeast, two pharmaceutical companies recently announced very positive news about a vaccine candidate.
Pfizer Inc. and Valneva SE announced on March 23, 2026, topline results from the Phase 3 VALOR "Vaccine Against Lyme for Outdoor Recreationists" clinical trial (NCT05477524) of its investigational 6-valent OspA-based Lyme disease vaccine candidate PF-07307405 (LB6V, formerly known as VLA15).
In the pre-specified analyses: Efficacy of 73.2% from 28 days post-dose 4 (season 2) in reducing the rate of confirmed Lyme disease cases compared to the placebo arm (95% CI 15.8, 93.5); Efficacy of 74.8% from 1-day post-dose 4 (season 2) in reducing the rate of confirmed Lyme disease cases compared to the placebo arm (95% CI 21.7, 93.9)
The press release stated, 'Given the clinically meaningful efficacy and the fact that the 95% confidence interval lower bound was above 20 in the second pre-specified analysis, Pfizer is confident in the vaccine's potential and is planning submissions to regulatory authorities.'
"Lyme disease can cause potentially serious consequences – where individuals and families face symptoms that can disrupt daily life, work, and long-term health – and there is currently no vaccine available," said Annaliesa Anderson, Ph.D., Senior Vice President and Chief Vaccines Officer, Pfizer. "The efficacy shown in the VALOR study of more than 70% is highly encouraging and creates confidence in the vaccine's potential to protect against this disease that can be debilitating."
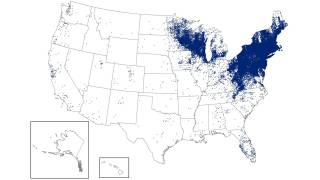
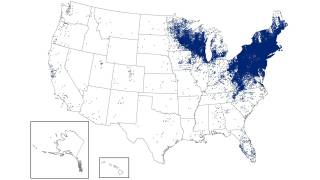
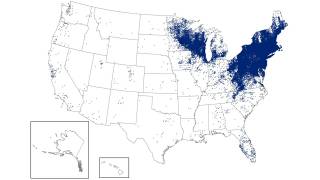
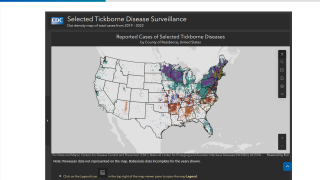
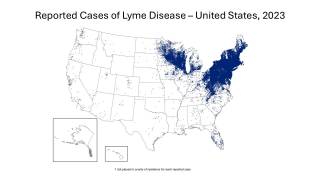
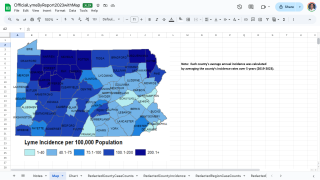
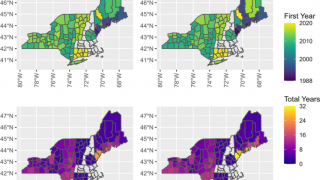
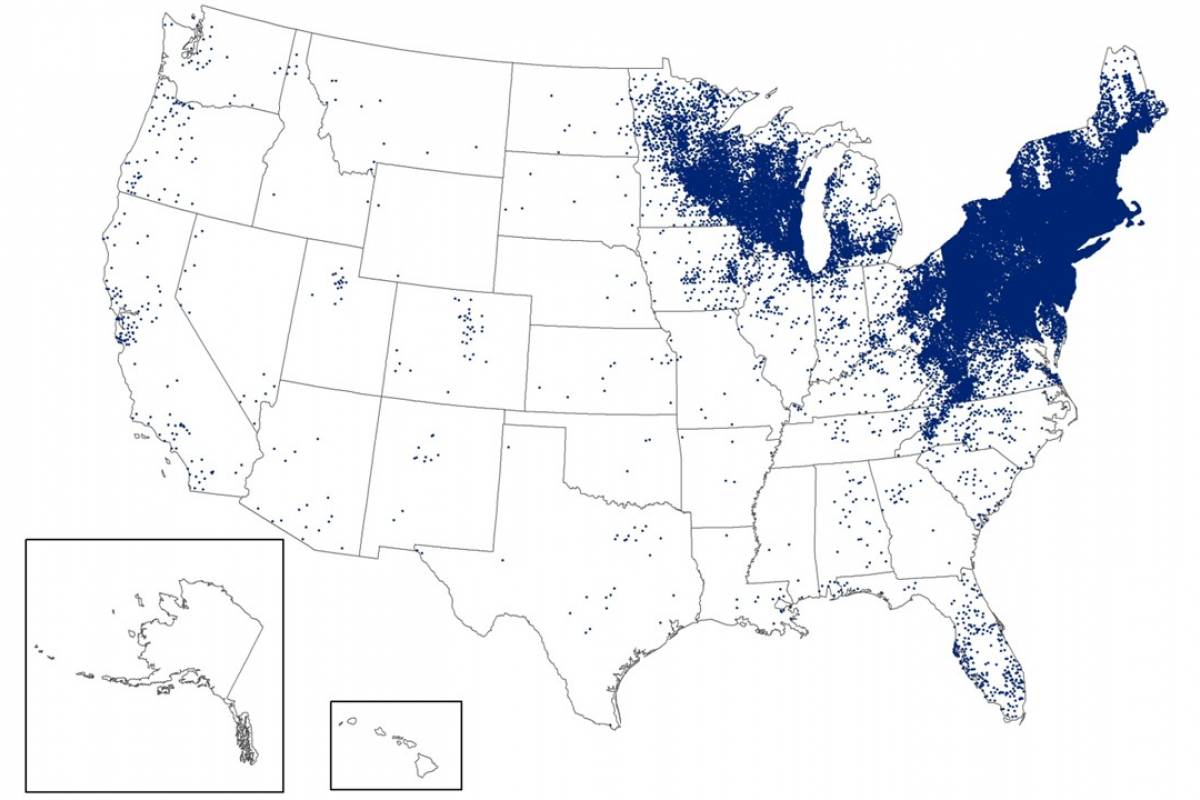
As of late March 2026, the U.S. CDC reports that the bacteria that cause Lyme disease are transmitted to humans by blacklegged (Ixodes) ticks. Infected ticks must be attached for over 24 hours to transmit the infection, so that prompt removal can prevent transmission. These ticks are most commonly found in forested areas of the northeastern, north-central, and mid-Atlantic states, as well as parts of the Pacific Coast.
The CDC reports that recent estimates using other methods suggest that approximately 476,000 people may be diagnosed and treated for Lyme disease each year in the United States. This number likely includes patients who are treated based on clinical suspicion but do not actually have Lyme disease.
Our Trust Standards: Medical Advisory Committee