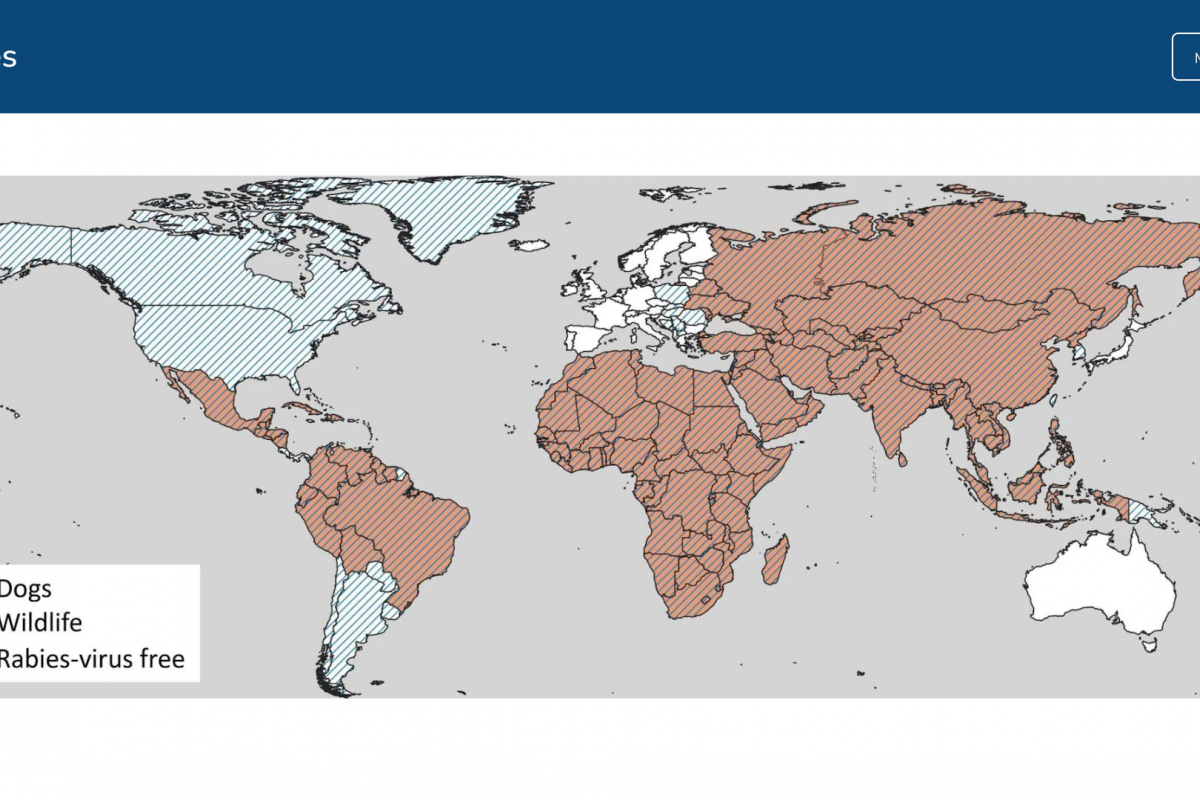
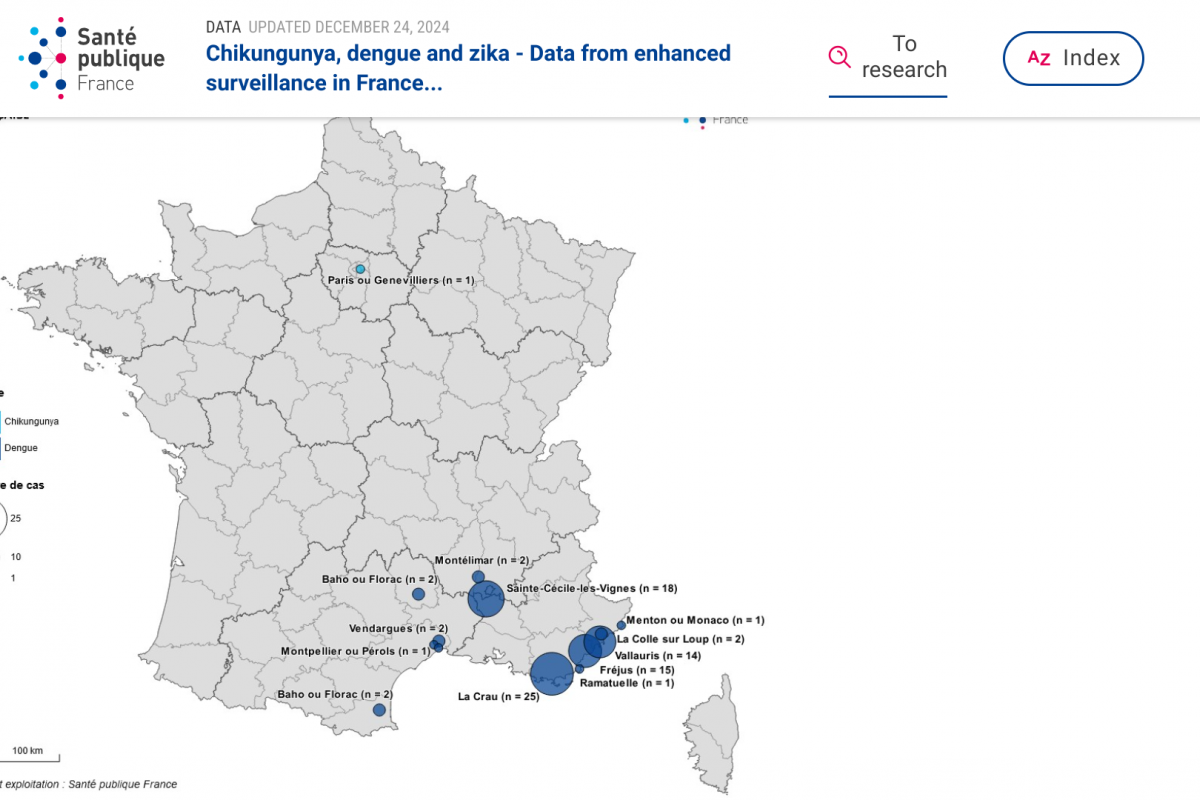
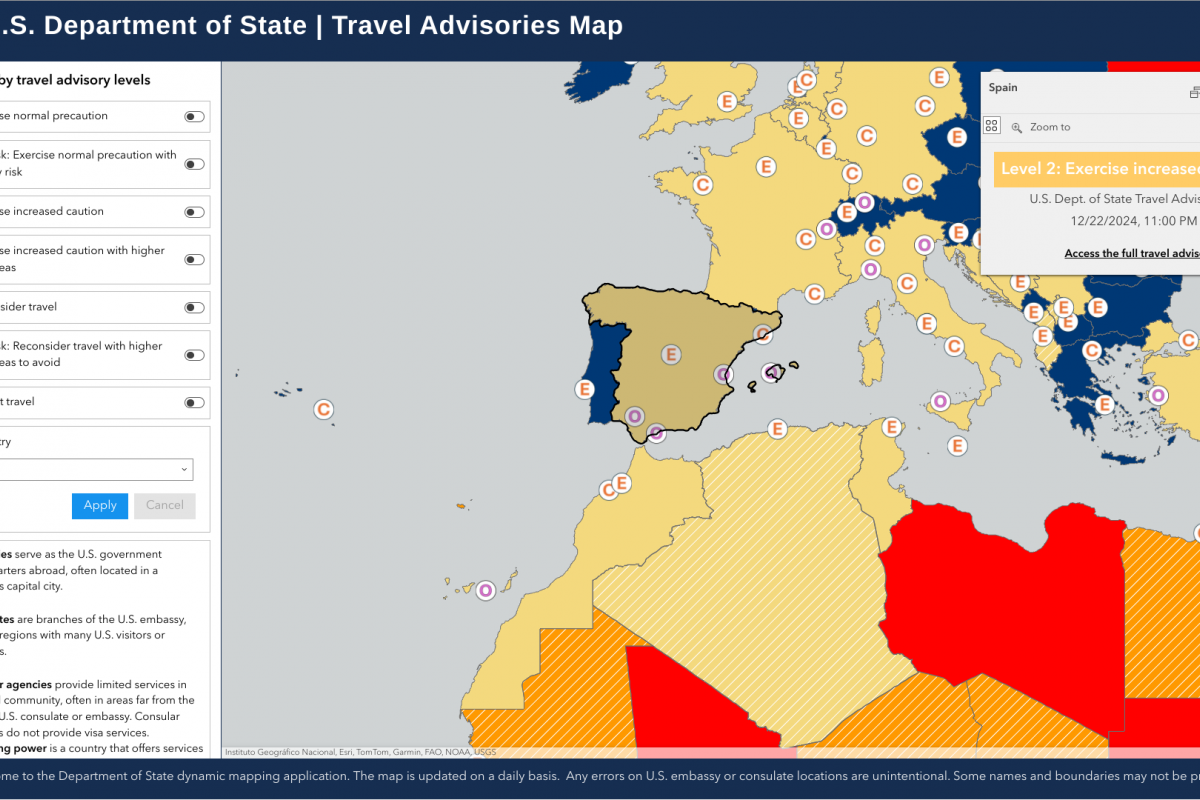
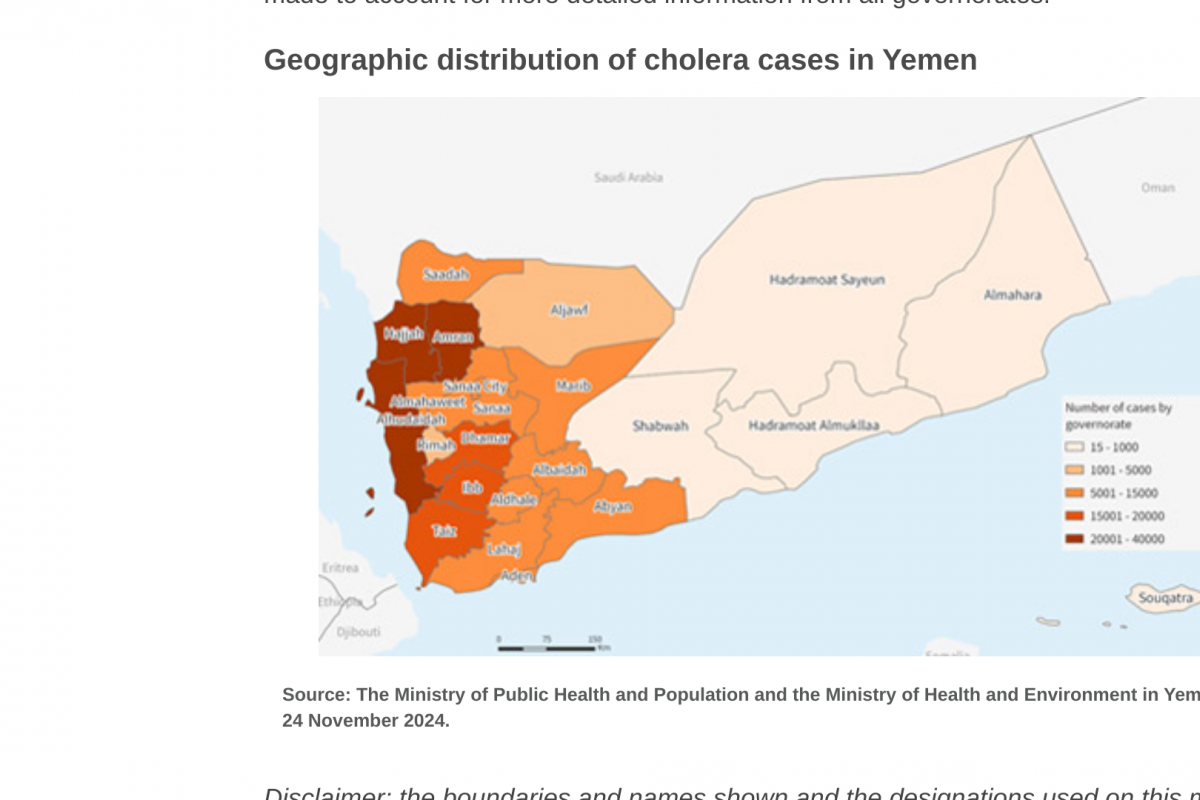
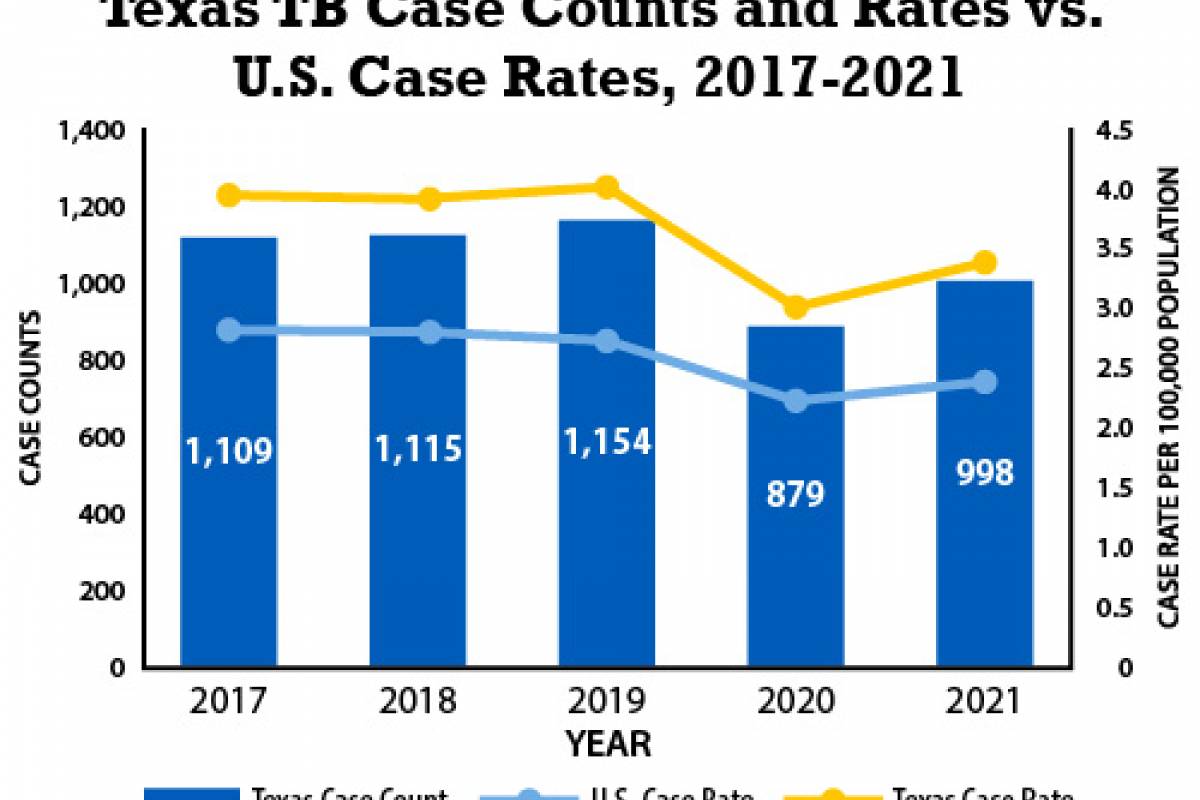
The Pan American Health Organization (PAHO) recently confirmed that influenza-like illnesses have increased in the North American subregion. Seasonal influenza (flu) became widespread in many sections of the United States in late December 2024.
According to the PAHO and numerical health agencies, most eligible people should get a flu shot that protects against the viruses causing infection.
The PAHO says there are four types of influenza viruses: A, B, C, and D.
As of December 25, 2024, various U.S. FDA-approved flu shots are available at local pharmacies, helping prevent severe influenza infections caused by these viruses.
The good news is pharmaceutical companies are developing vaccines that provide broad-spectrum protection against these every-mutating viruses.
A study published on December 11, 2024, in the journal MDPI, reported in a Phase 2a, double-blind, placebo-controlled study, OVX836, a nucleoprotein (NP)-based candidate vaccine, previously showed a good safety profile, a robust immune response (both humoral and cellular) and a preliminary signal of protection of 84% against confirmed symptomatic influenza after a single intramuscular dose of 180 µg, 300 µg or 480 µg.
Furthermore, T-cell responses were highly cross-reactive against various influenza A strains, both seasonal and highly pathogenic avian strains.
Last month, Osivax announced its ongoing efforts to prepare this vaccine candidate for marketing.
On November 11, 2024, the first participant was vaccinated in a Phase 2a clinical trial (NCT06582277) evaluating OVX836 as a booster in participants vaccinated three to five years ago in earlier Osivax vaccine studies.
The topline results from this trial are expected by the end of 2025.
“This milestone is a significant step forward in our mission to develop a truly broad-spectrum, lasting flu vaccine capable of addressing the ever-evolving threat of influenza. By studying the effects of a booster dose, we aim to deepen our understanding of OVX836’s potential to provide robust and sustained immune protection,” said Dr. Nicola Groth, CMO of Osivax, in a press release.
“Osivax is committed to leveraging innovative science to develop vaccines that protect individuals and help reduce the global healthcare burden associated with seasonal flu epidemics and potential pandemics.”
Osivax is a clinical-stage biopharmaceutical company that aims to develop a pan-respiratory virus vaccine that can prevent all strains of influenza in one shot. The company also intends to expand into other infectious disease indications through combinations and collaborations worldwide.