A shipment of 11,200 vaccine doses, donated by the United States of America, has been shipped to Abuja, Nigeria, to curtail the ongoing clade 1 mpox outbreak.
This shipment, announced on December 20, 2024, follows agreements signed in November by Gavi, the Vaccine Alliance, to facilitate the donation of 305,000 mpox vaccine doses to support the global and continental response.
In September 2024, the U.S. announced its intention to donate up to 1 million JYNNEOS® (MVA-BN®) doses to support the mpox emergency.
Assistant Secretary for Preparedness and Response (ASPR) Dawn O'Connell commented in a press release, "Viruses don't respect borders, and both international and domestic mpox coordination remains a top priority for ASPR."
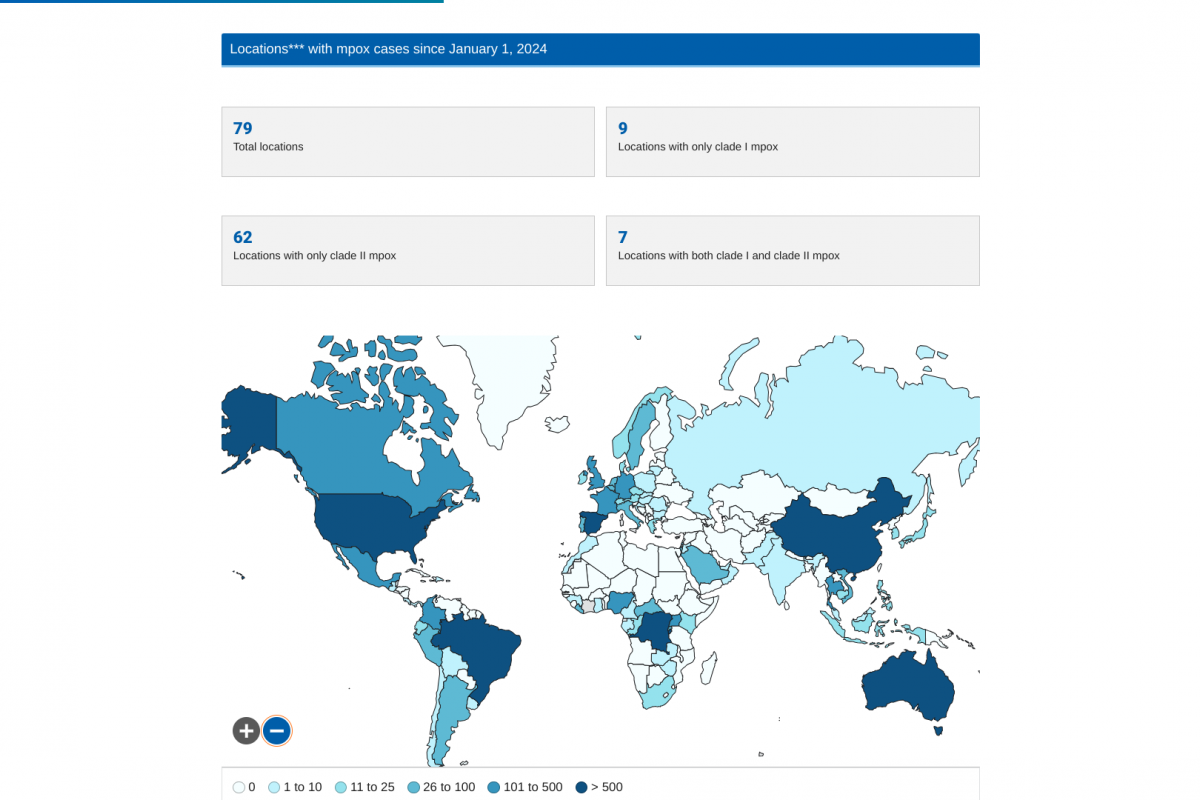
Two types of the virus cause mpox, clade I and clade II. Both types spread the same way and can be prevented using innovative vaccines.
The first case of clade I mpox in the U.S. was detected in November 2024 following the patient's travel to an affected area. No additional cases were reported.
The initial case of clade IIb mpox in the U.S. (Boston) was in May 2022. According to the U.S. CDC, the ongoing global outbreak of clade II mpox has caused more than 100,000 cases in 122 countries and continues in the U.S.
Bavarian Nordic's JYNNEOS® two-dose vaccine is based on a live, attenuated vaccinia virus, Modified Vaccinia Ankara, and is commercially available in the U.S.