The U.S. NIH recently announced the antiviral drug tecovirimat, known commercially as TPOXX®, did not reduce the time to lesion resolution or have an effect on pain among adults with mild to moderate clade II mpox and a low risk of developing severe disease.
A planned interim analysis at 75% of the current study's target enrollment showed no difference in the time to lesion resolution between participants treated with tecovirimat compared with those who received a placebo.
This finding is based on an interim data analysis from an international clinical trial called the Study of Tecovirimat for Mpox (STOMP).
Considering these definitive findings, the study's Data Safety and Monitoring Board recommended stopping further enrollment of participants who were being randomized to tecovirimat or placebo.
At the Board's request, an additional assessment was performed. Based on the study design and available data, there was less than a 1% chance that the study would show that tecovirimat would be effective if it were to complete enrollment and follow-up.
"The initial STOMP findings provide valuable insight to inform clade II mpox medical countermeasures and underscore the critical importance of conducting well-designed randomized clinical trials during infectious disease outbreaks," said NIAID Director Jeanne Marrazzo, M.D., M.P.H., in a media release on December 10, 2024.
"Before 2022, no treatment candidate had been studied in people with mpox, and this trial is a critical step in our systematic evaluation of existing antivirals like tecovirimat while pursuing novel antivirals and antibody-based mpox therapeutics."
The Food and Drug Administration initially approved SIGA Technologies, Inc.'s Tecovirimat to treat smallpox.
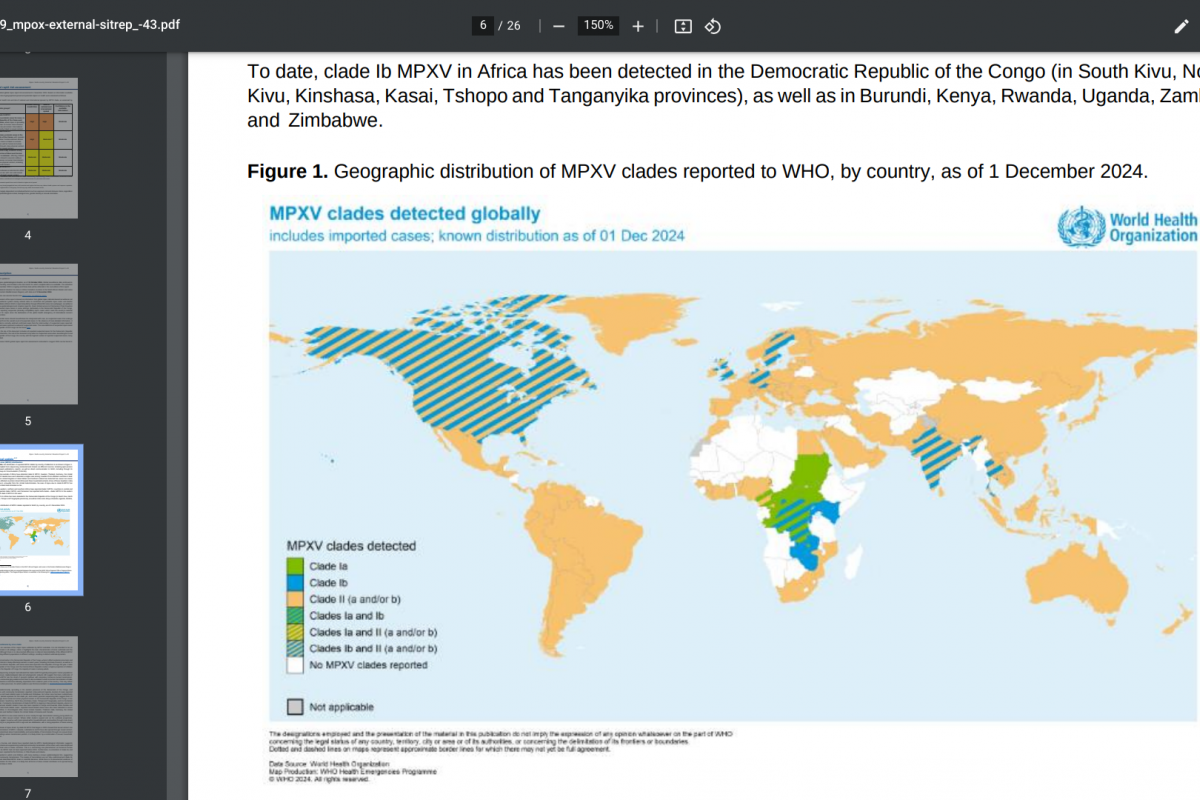
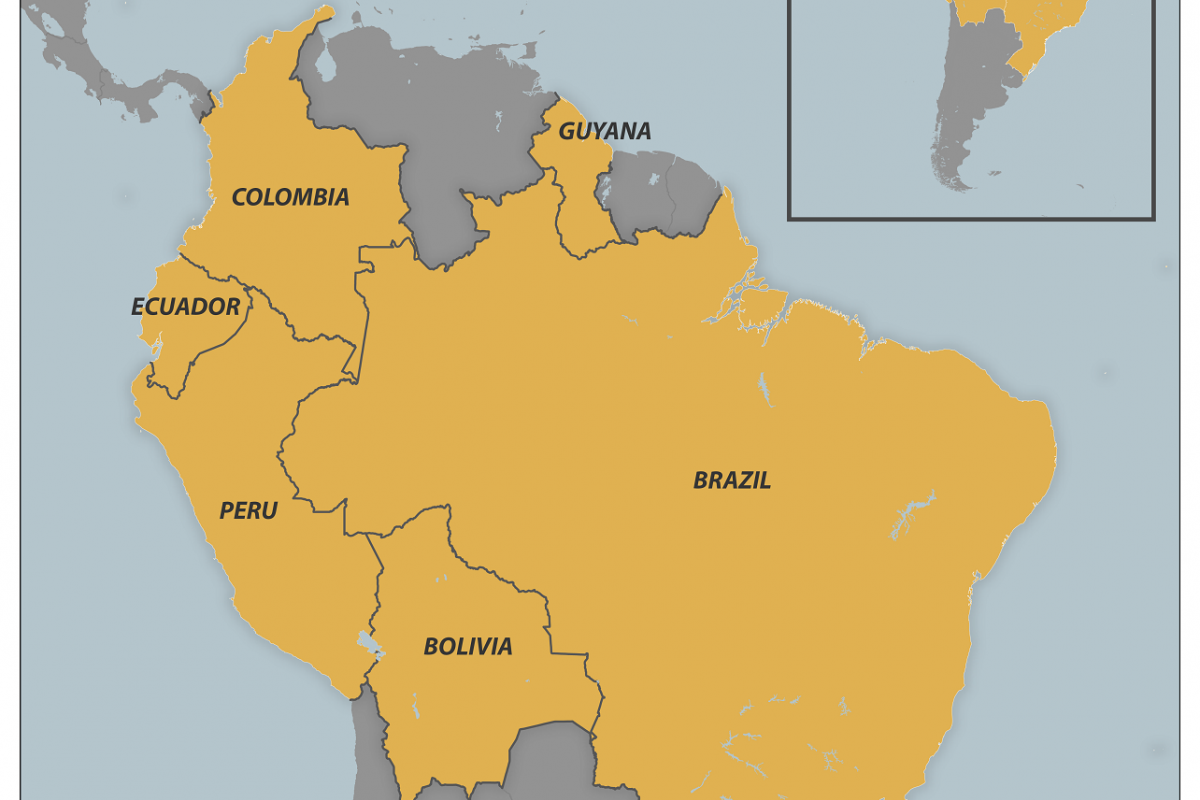
Mpox is caused by a virus that spreads mainly through close contact. Two virus types, clades I and II, have been identified and are historically present in Central and West Africa.
A clade II subtype virus caused a global mpox outbreak in 2022, and the virus continues to circulate at low levels.
In 2024, the World Health Organization declared a Clade I outbreak in Central and East African countries a public health emergency of international concern.
Travel-related cases of clade I mpox have been reported internationally, and the first reported case in the United States was diagnosed on November 15, 2024.
The U.S. CDC has issued a Level 2 travel advisory regarding the Mpox outbreak.