In Europe, dengue viruses, transmitted by Aedes albopictus mosquitoes, are primarily associated with infections acquired in endemic countries.
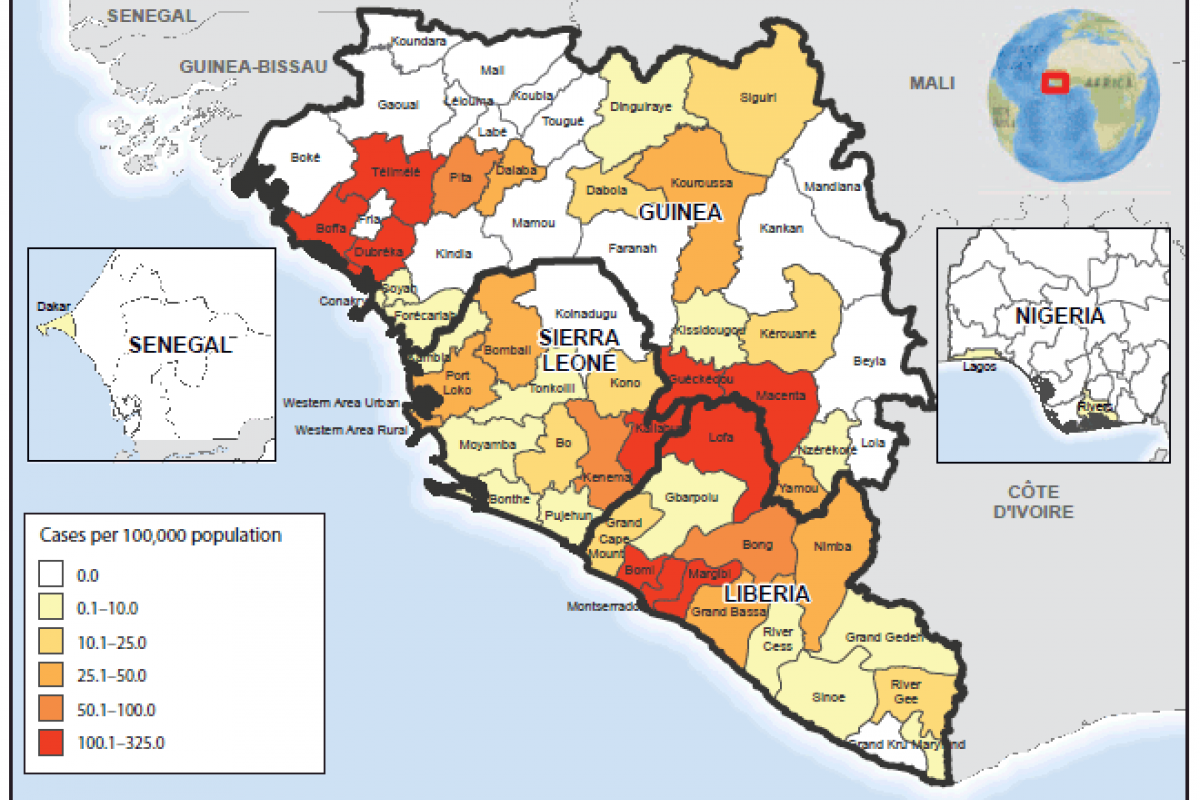
However, autochthonous (locally acquired) dengue cases have surged in the past five years, with significant reports from Spain, Croatia, France, and Italy. In 2023, Italy reported its highest-ever number of locally transmitted dengue virus cases.
The year 2023 was exceptional, not just due to the total number of reported cases but also for the uncommon co-occurrence of a few spatially disjointed autochthonous transmission chains of more than one DENV serotype, wrote researchers in an article published by the journal Nature on December 5, 2024.
The Italian National Public Health Authority reported 213 locally acquired dengue cases in 2024, a significant increase from the 82 confirmed cases last year.
Specifically, Italy's dengue outbreaks have become regionalized as infected mosquitoes spread the virus.
As of October 28, 2024, Eurosureliiance reported 138 confirmed and 61 probable cases of DENV-2 in Fano, a small coastal city in Italy's Marche Region.
With the holiday travel season fast approaching, Italy is anticipating additional dengue cases to be confirmed in 2024.