Innovative Test PreQualified for the World's Most Infectious Diseases
The World Health Organization (WHO) today announced it has granted prequalification to the molecular diagnostic test for tuberculosis (TB) called Xpert® MTB/RIF Ultra.
As of December 5, 2024, this is the first test for TB diagnosis and antibiotic susceptibility testing that meets WHO's prequalification standards.
This nucleic acid amplification test (NAAT) Xpert® MTB/RIF Ultra detects the genetic material of Mycobacterium tuberculosis, the bacterium that causes TB, in sputum samples and provides accurate results within hours.
Simultaneously, the test identifies mutations associated with rifampicin resistance, a key indicator of multidrug-resistant TB.
"This first prequalification of a diagnostic test for tuberculosis marks a critical milestone in WHO's efforts to support countries in scaling up and accelerating access to high-quality TB assays that meet both WHO recommendations and its stringent quality, safety, and performance standards," said Dr. Yukiko Nakatani, WHO Assistant Director-General for Access to Medicines and Health Products, in a press release.
"It underscores the importance of such groundbreaking diagnostic tools in addressing one of the world's deadliest infectious diseases."
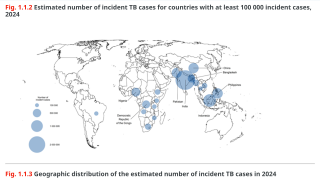
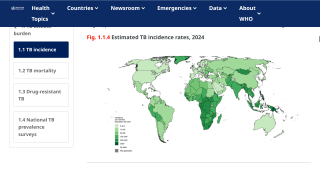
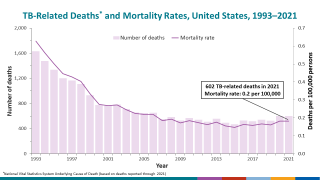
Tuberculosis outbreaks are one of the world's leading infectious disease killers, causing over a million deaths annually and imposing immense socioeconomic burdens, especially in low- and middle-income countries. Accurate and early detection of TB, especially drug-resistant strains, remains a critical and challenging global health priority.
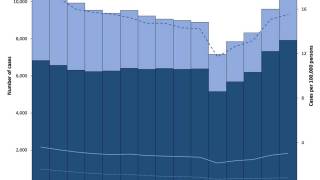
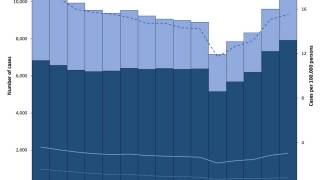
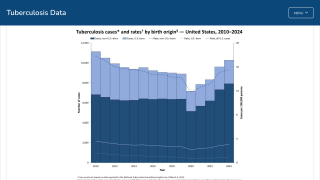
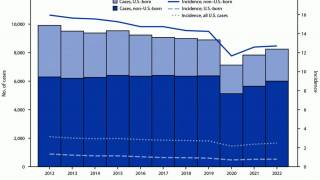
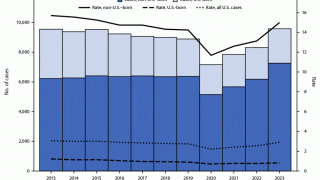
In the United States, TB cases have been increasing over the past few years.
Additionally, TB is a vaccine-preventable disease, with over 10 BCG vaccines available worldwide.
Our Trust Standards: Medical Advisory Committee