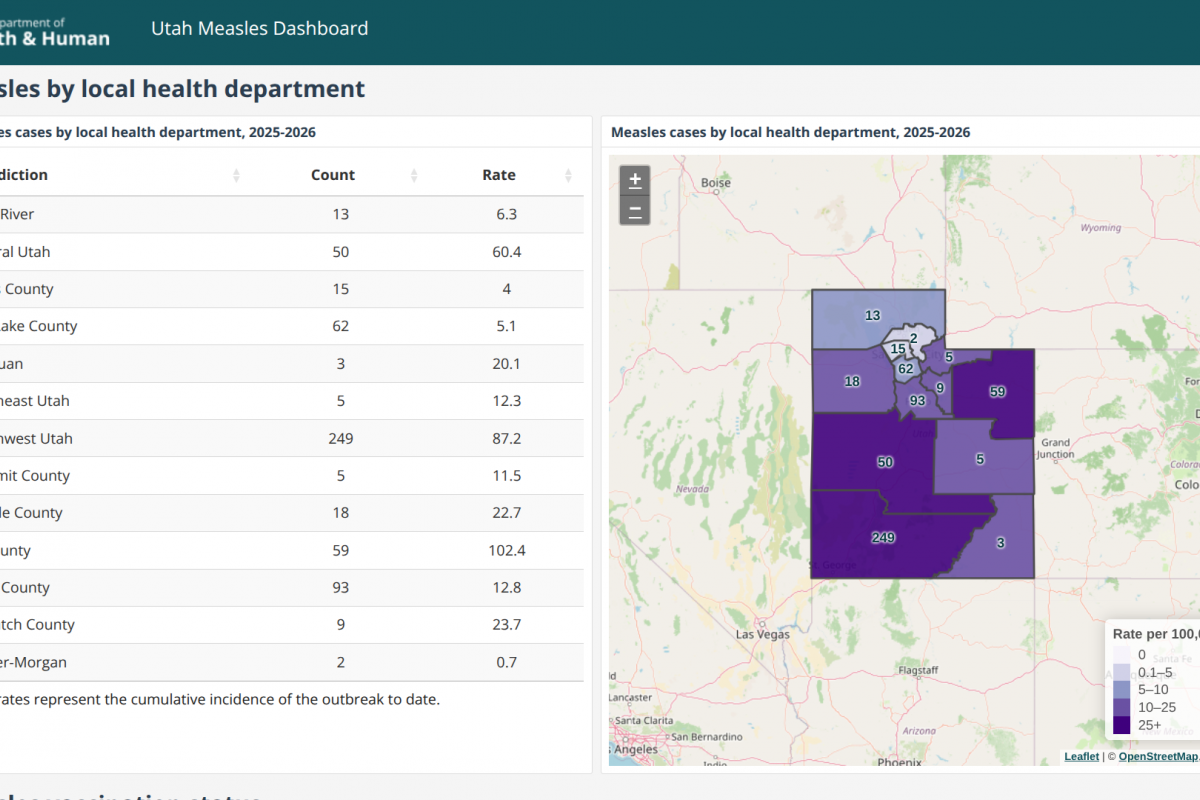
Philippine health authorities announced (NO.2026-0347) that they have dismantled an illegal manufacturing and distribution ring producing counterfeit anti-rabies vaccines during a coordinated raid in Taguig City.
The Philippine Food and Drug Administration (FDA), along with other agencies, executed a court-issued search warrant following weeks of surveillance and intelligence gathering. The raid uncovered an unlicensed facility involved in the falsification and repackaging of anti-rabies vaccines.
Items seized during the operation included counterfeit sticker labels, hologram adhesive tapes, vaccine cartons, unlabeled vials, and diluents intended to misrepresent the products as genuine.
FDA officials described the operation as a significant victory against the distribution of counterfeit medicines. Local officials believe the syndicate smuggled fake vaccines and concealed them among legitimate supplies to avoid detection.
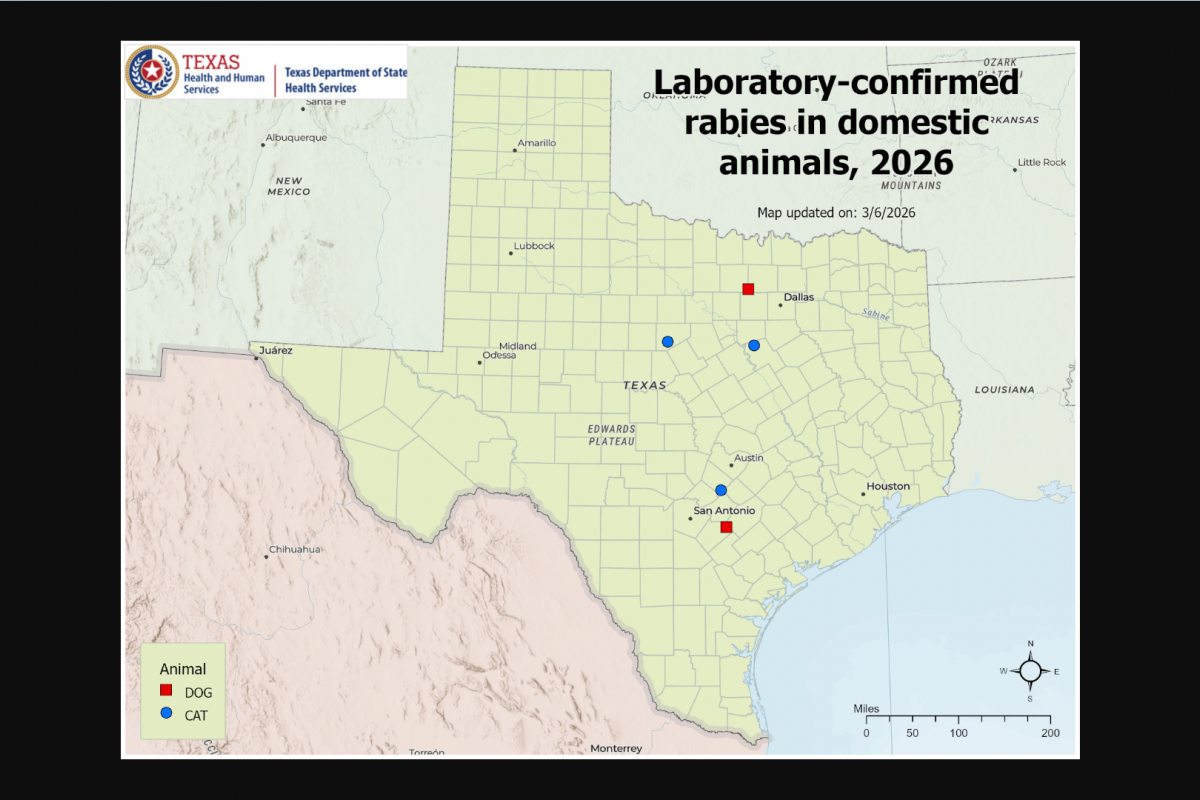
Counterfeit or substandard rabies vaccines can fail to provide protection, leaving recipients vulnerable to rabies — a disease that is nearly 100% fatal once symptoms develop.
In the United States, rabies vaccines are strictly regulated by the U.S. FDA. The two primary human rabies vaccines currently approved are Imovax Rabies (human diploid cell vaccine) and RabAvert (purified chick embryo cell vaccine).
According to the U.S. Centers for Disease Control and Prevention (CDC), approximately 100,000 Americans receive rabies post-exposure prophylaxis (PEP) each year following potential exposure to the virus. PEP typically consists of wound care, human rabies immune globulin, and a four-dose vaccine series administered for previously unvaccinated individuals.
The CDC says an additional 1.4 million Americans seek medical evaluation annually for possible rabies exposures.
In the U.S., most rabies bites originate from bats.