As vacationers plan their winter 2025 holiday trip to Florida's beautiful beaches to enjoy the warm weather, taking steps to prevent mosquito bites remains essential. In Florida, 14 species of Anopheles mosquitoes have been found to transmit diseases to humans.
As of early January 2025, mosquito-borne diseases, such as dengue fever, have been reported again in Florida.
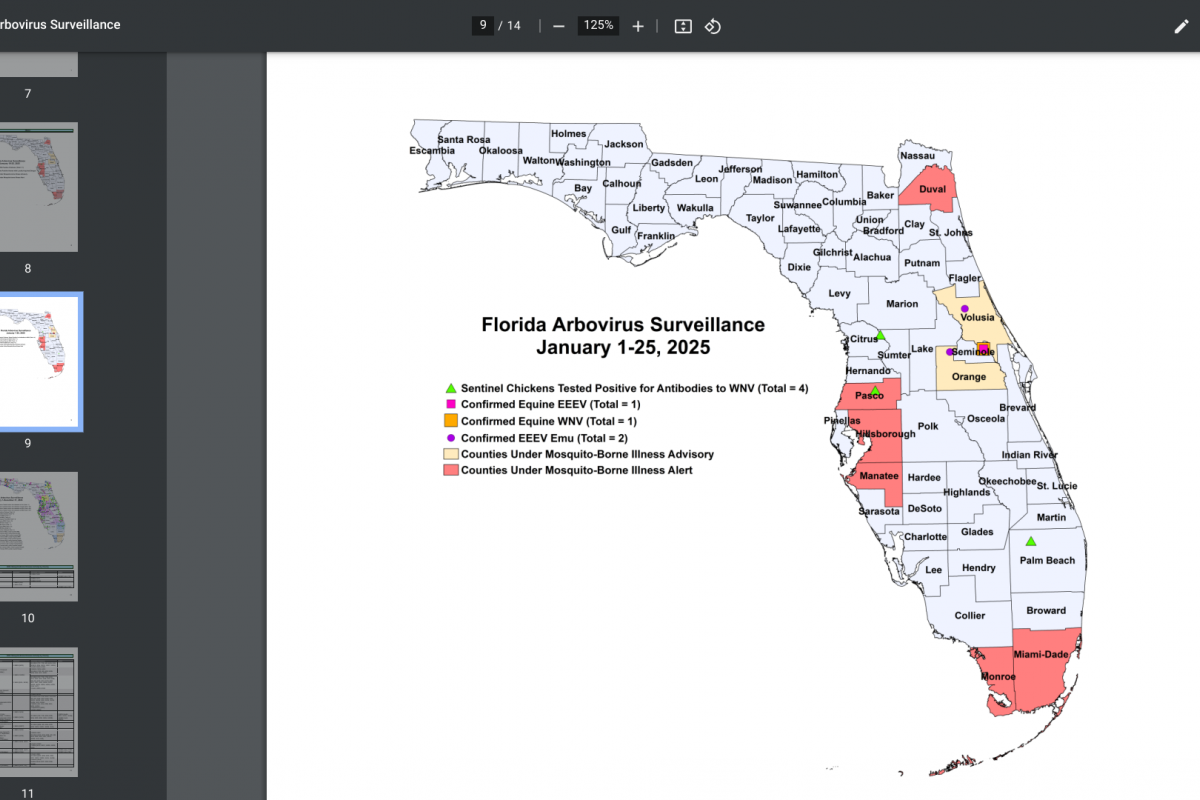
The Florida Department of Health (FDH) updated its Vaccine-Preventable Diseases Surveillance Report on January 25, 2025, confirming twenty-two cases of dengue were reported among persons who had international travel, and one locally acquired dengue case was reported.
Last year, 999 travel-associated dengue cases were reported, mainly among Brazil, Cuba (567), and Puerto Rico visitors.
Furthermore, 91 locally acquired dengue cases were reported from ten counties in 2024, led by Miami-Dade (50).
While dengue preventive vaccines remain unavailable in the United States, FDH and the U.S. CDC encourage all visitors to dengue-endemic areas to avoid mosquito bites. This CDC advice is particularly relevant for pregnant women, as the dengue virus has been detected in unborn infants.