Over the past two years, the U.S. Centers for Disease Control and Prevention (CDC) has issued Travel Health Advisories focused on measles outbreaks.
These advisories highlight where there is an active health risk when people visit the highlighted countries.
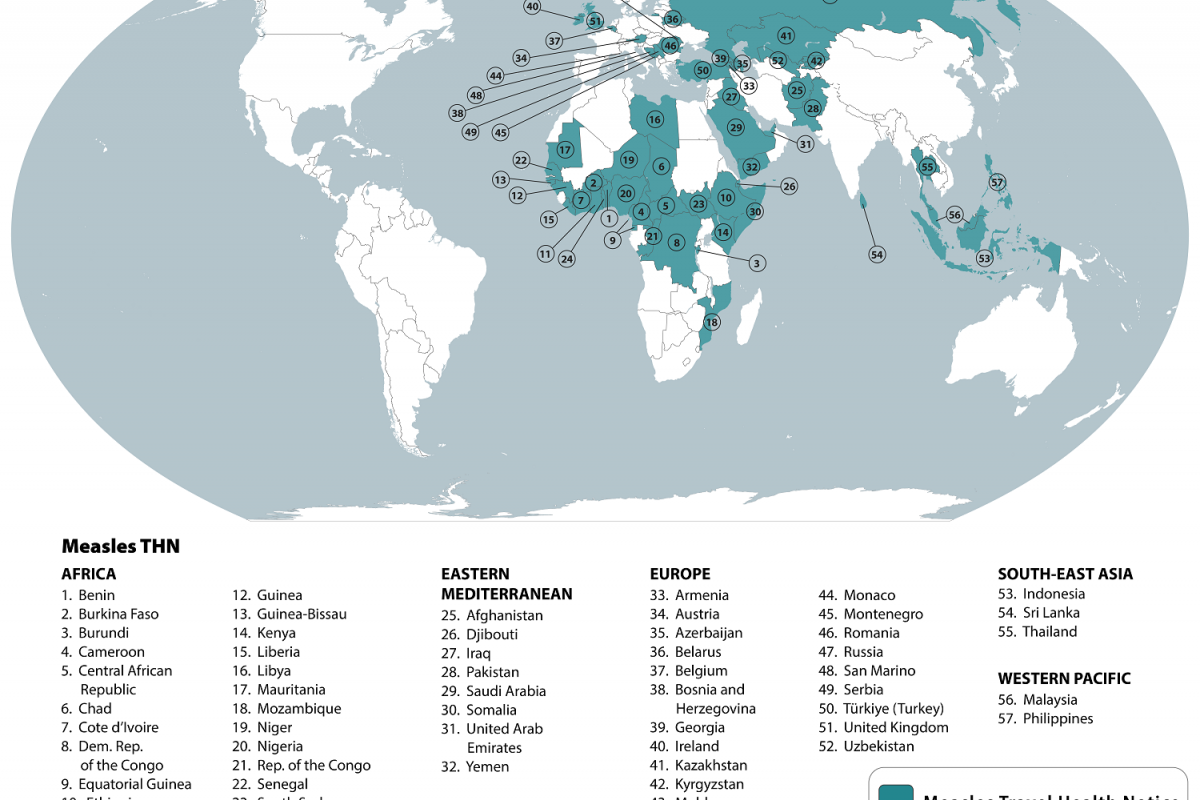
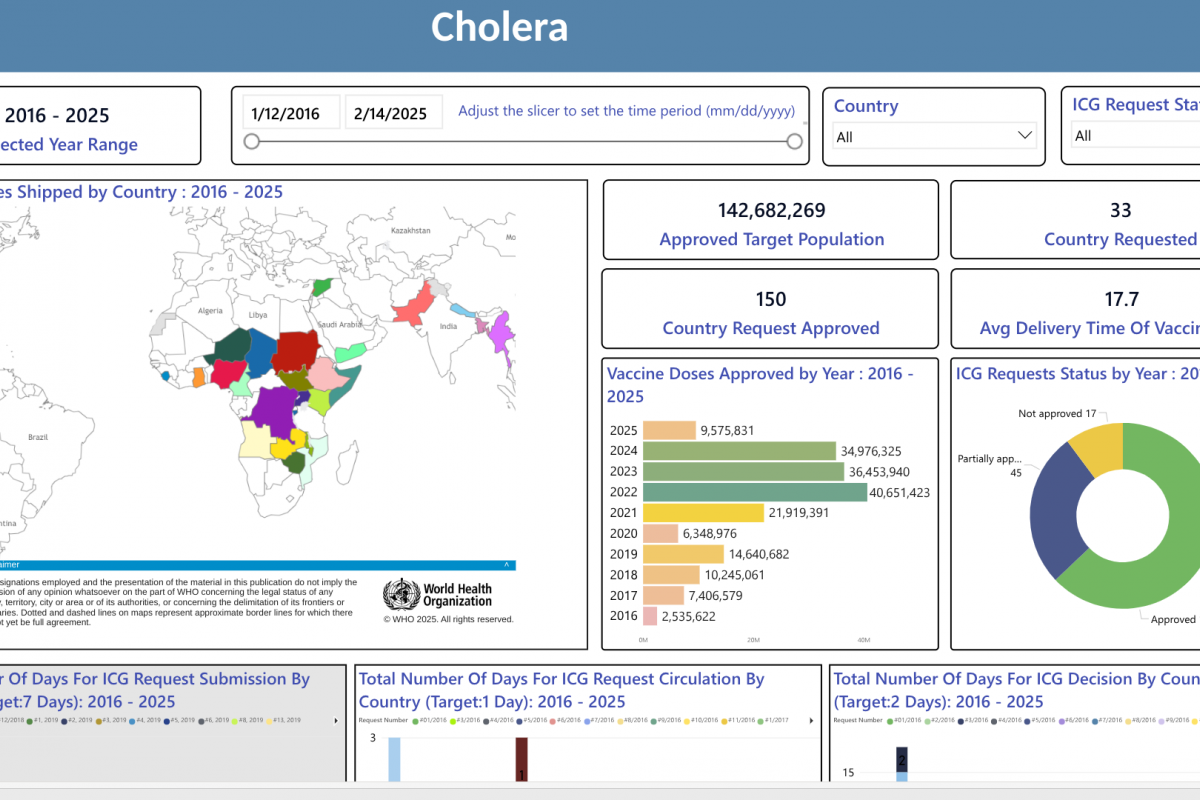
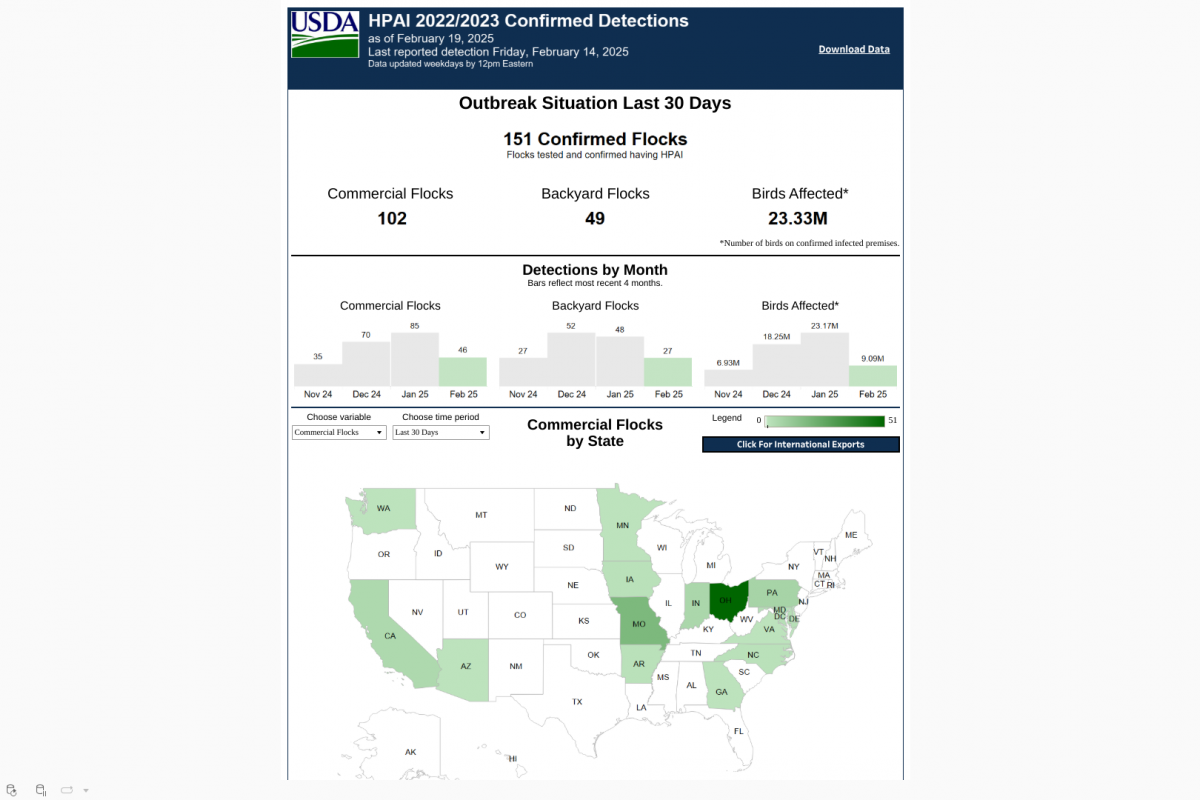
On February 21, 2025, the CDC reissued a Level 1, Practice Usual Precautions, alert for 57 countries. This CDC list does not integrate the Region of the Americas, with numerous countries reporting 537 measles outbreaks this year.
This is an essential advisory since most measles cases imported into the United States occur in unvaccinated U.S. residents who become infected during international travel.
The CDC recently confirmed 93 measles cases in eight jurisdictions, which may be under-reporting active outbreaks in New Mexico and Texas.
The CDC says to reduce measles cases in the U.S., 'All international travelers should be fully vaccinated against measles with the measles-mumps-rubella (MMR) vaccine, including an early dose for infants.'
In 2025, MMR vaccinations are offered at most clinics and pharmacies in the U.S.