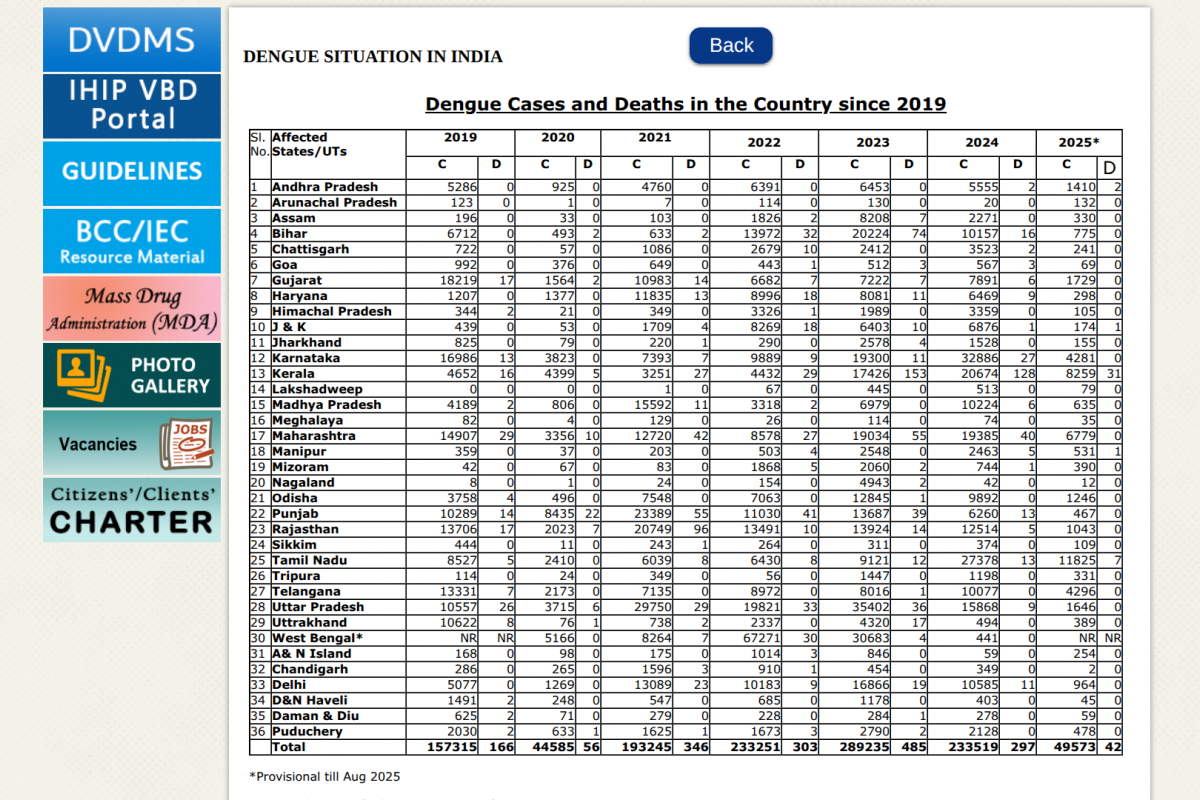
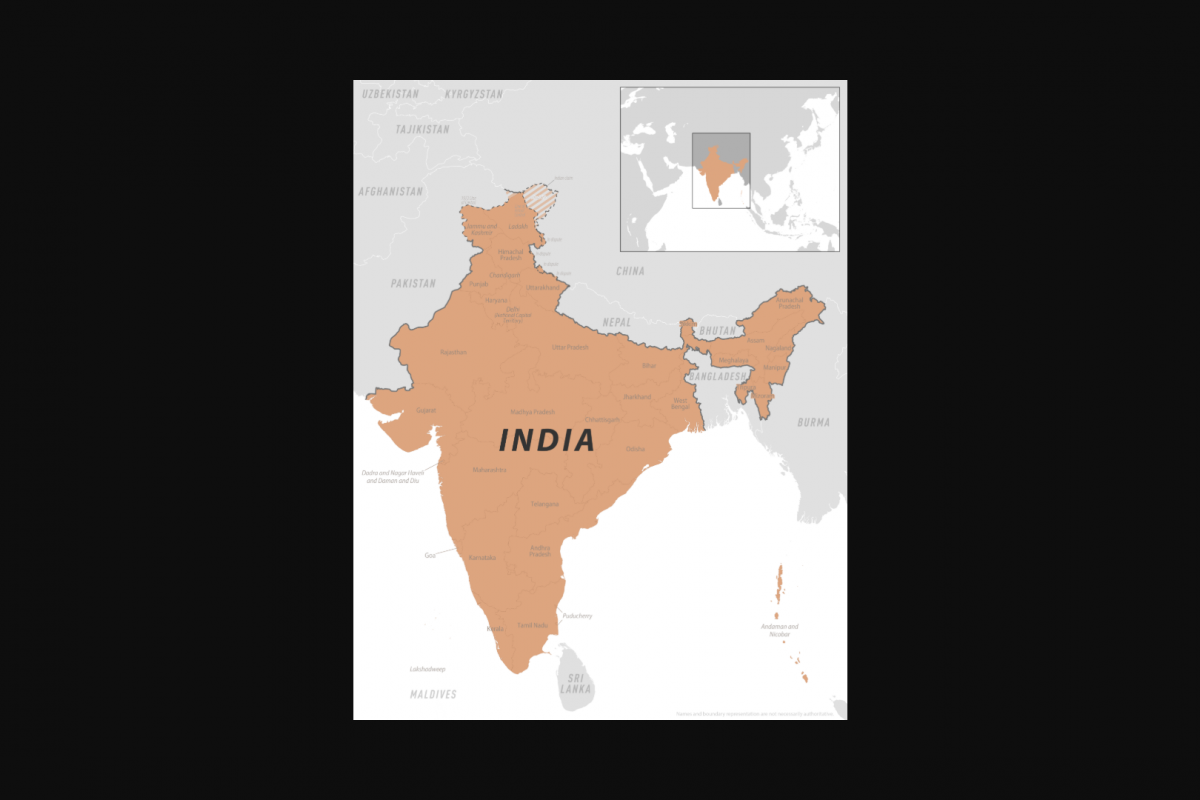
Throughout 2025, India has reported an increase in dengue cases across most regions. With the new year fast approaching, the National Centre for Vector Borne Disease Control (NCVBDC) data indicates a difficult season for this mosquito-transmitted disease in 2026.
The NCVBDC data indicate that, in October 2025, a total of 22,659 dengue cases were reported in India, a 21% increase compared to September 2025 (18,803). Tamil Nadu is the unfortunate leader in 2025.
And as of December 2025, over 49,000 dengue cases had been reported.
When the U.S. CDC last updated its Level 1 - Practice Usual Precautions, the Global Travel Health Notice focused on dengue outbreaks; it did not include a warning for India. However, the CDC says Travelers visiting dengue outbreak countries may be at increased risk in 2025.
Furthermore, the CDC has identified a higher-than-expected number of dengue cases among U.S. travelers returning from those countries, including those arriving in Florida.
While the CDC does not recommend the second-generation dengue vaccine known as Qdenga, various countries do offer it in December 2025.