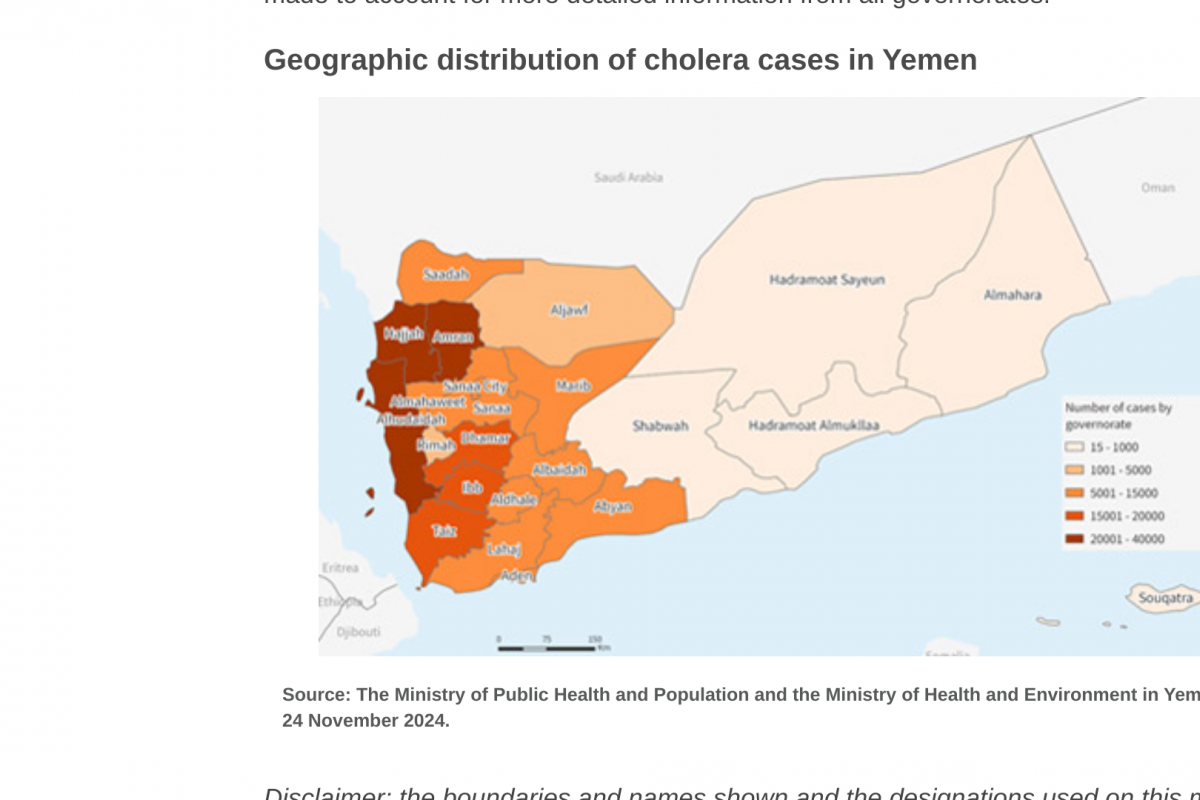
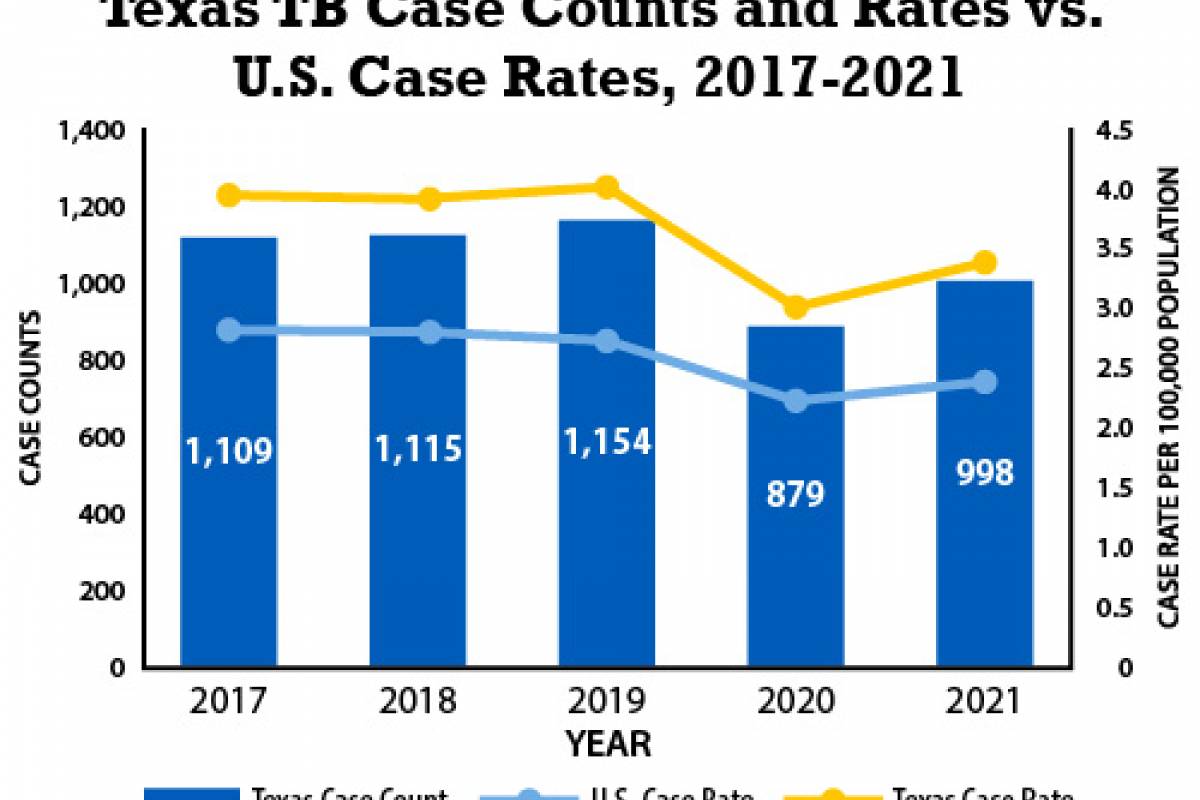
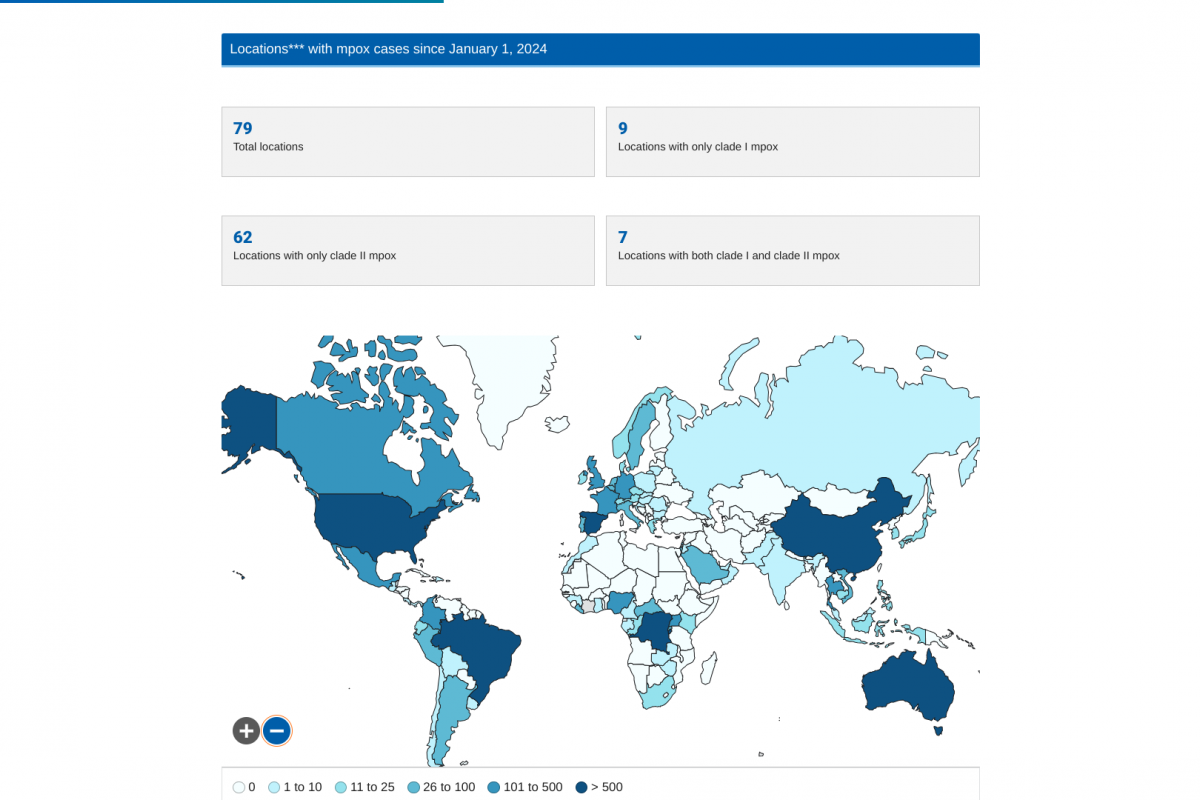
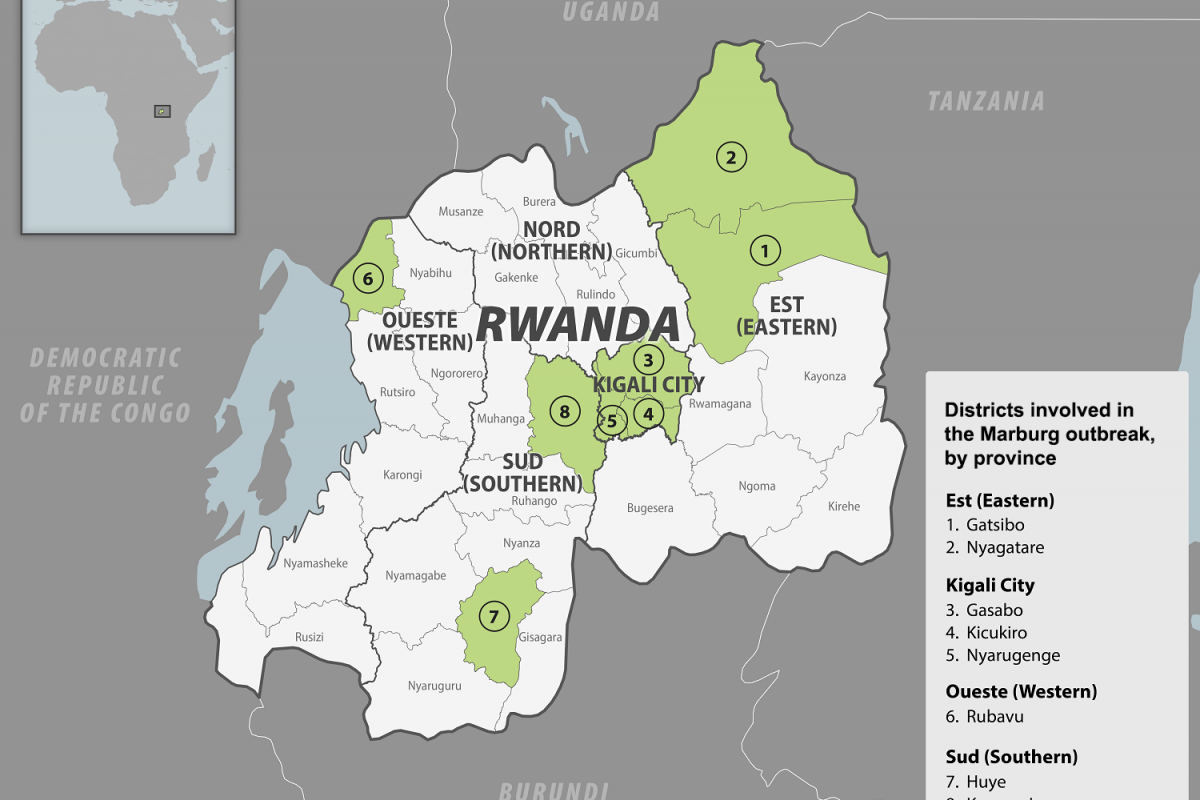
Despite decades of public health vaccination programs, invasive pneumococcal disease (IPD) continues to cause substantial disease burden, primarily due to Streptococcus pneumoniae serotypes not included in pneumococcal conjugate vaccines (PCV).
Various pharmaceutical companies say next-generation PCVs can extend vaccine coverage of disease-causing serotypes.
To accelerate solutions addressing this health need, Sanofi and SK bioscience announced on December 23, 2024, that they entered into a new collaboration in pneumococcal vaccines with an expanded agreement to develop, license and commercialize next-generation PCVs for pediatric and adult populations.
This expansion builds on the companies’ existing collaboration to develop and commercialize a PCV21 pediatric vaccine, for which the phase 3 clinical program commenced last week. This vaccine candidate is the first-ever PCV containing more than 20 serotypes to enter a phase 3 clinical study in infants and toddlers.
The PCV21 phase 3 program, based on positive phase 2 results communicated in 2023, will include infants, toddlers, young children, and adolescents across multiple geographies, including the US, Europe, Australia, Asia, and Latin America.
Thomas Triomphe, Executive Vice President of Vaccines at Sanofi, commented in a press release on December 23, 2024, “Given the vast unmet public health needs in IPD, we’re delighted to expand this collaboration and continue our pursuit of innovative work in PCV. Our collaboration leverages SK bioscience’s capabilities and Sanofi’s expertise in developing and bringing innovative vaccines to people worldwide to reduce the global impact of pneumococcal disease.”
The World Health Organization says pneumococcal vaccinations continue to prevent pneumococcal disease. While vaccines can not prevent every kind of community-acquired pneumonia, they work against the most common bacteria types.