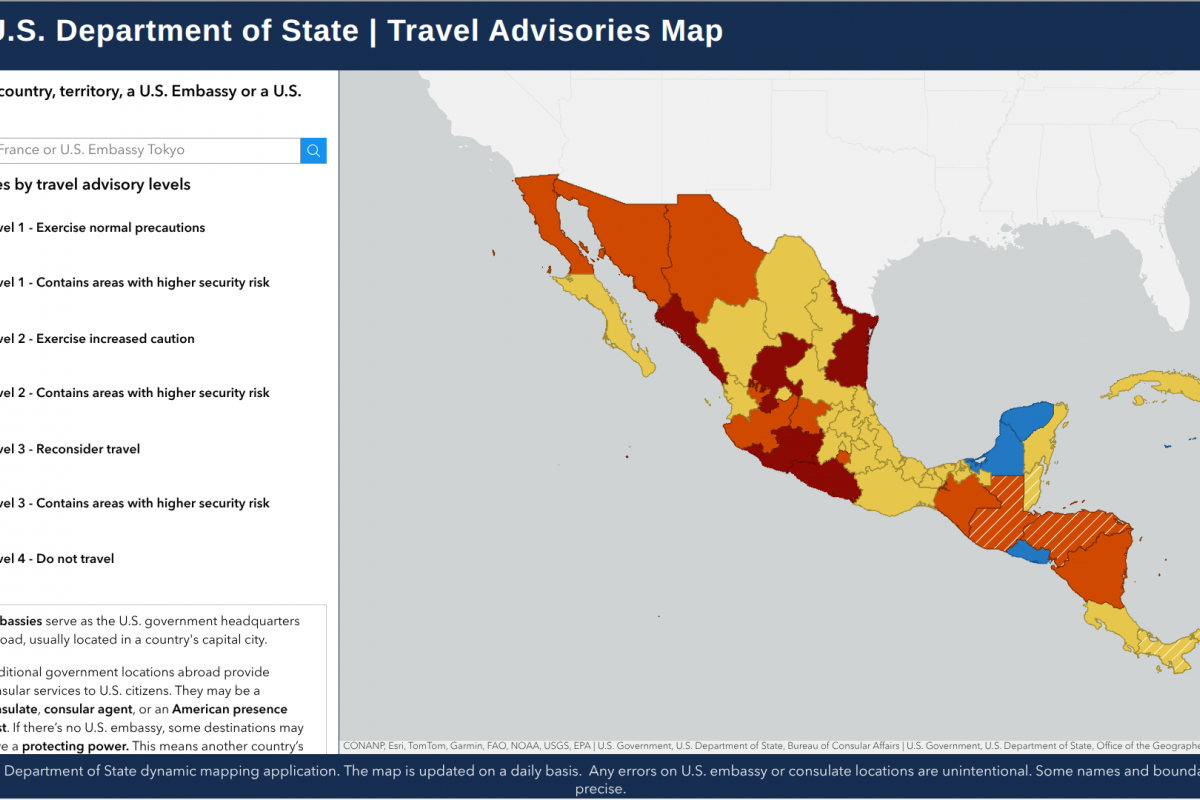
According to the U.S. Embassy and Consulates in Mexico, due to ongoing security operations, road blockages, and criminal activity, U.S. citizens in several locations in Mexico are advised to shelter in place.
As of February 23, 2026, Security Alert – Update 3 says U.S. government staff in Guadalajara (Jalisco), Puerto Vallarta (Jalisco/Nayarit), Ciudad Guzmán (Jalisco), Cancun (Quintana Roo), Playa del Carmen (Quintana Roo), Cozumel (Quintana Roo), Reynosa (Tamaulipas), Tijuana (Baja California), and Michoacán will shelter in place and work remotely on February 23.
Furthermore, U.S. citizens are encouraged to do the same.
Additionally, all U.S. government staff at the Consulate General in Monterrey have been instructed to remain within the Monterrey metropolitan area and advised not to travel to Mazatlán until Wednesday, February 25, 2026.
Regarding air travel, roadblocks have impacted airline operations, with most domestic and international flights cancelled in both Guadalajara and Puerto Vallarta.
All ride shares are suspended in Puerto Vallarta. Some businesses have suspended operations. And Mexican authorities announced that operations on toll roads in Puebla, Guerrero, Tamaulipas, Nayarit, San Luis Potosi, Tijuana, Queretaro, Veracruz, and Mazatlan will be temporarily curtailed due to blockades that have affected motorists.
These are a few of the actions the U.S. Department of States says people should take: Seek shelter and minimize unnecessary movements, Avoid areas around law enforcement activity, Check @CAPUFE on X for the status of road closures, Monitor local media for updates, Follow the directions of local authorities and in case of emergency, call 911, and Keep family and friends advised of your location and well-being via phone, text, and social media.
Seperately, Texas Governor Greg Abbott urged Texans currently in Mexico, as well as those with family members or friends traveling there, to closely follow warnings from the U.S. Department of State. "Texans' safety is my top priority, whether they are here at home or traveling abroad," said Governor Abbott in a press release.
"I urge all Texans in Mexico to follow the guidance of U.S. officials, stay alert, and ensure they are in contact with the U.S. Embassy or nearest U.S. Consulate. Situations on the ground can change quickly, and staying informed can save lives," added Abbott.
Since August 12, 2025, the State Department has issued Level 4 - Do not travel notices for several Mexican states.
Currently, the U.S. government recommends that you visit your health provider at least 1 month before traveling to Mexico to update your vaccinations.