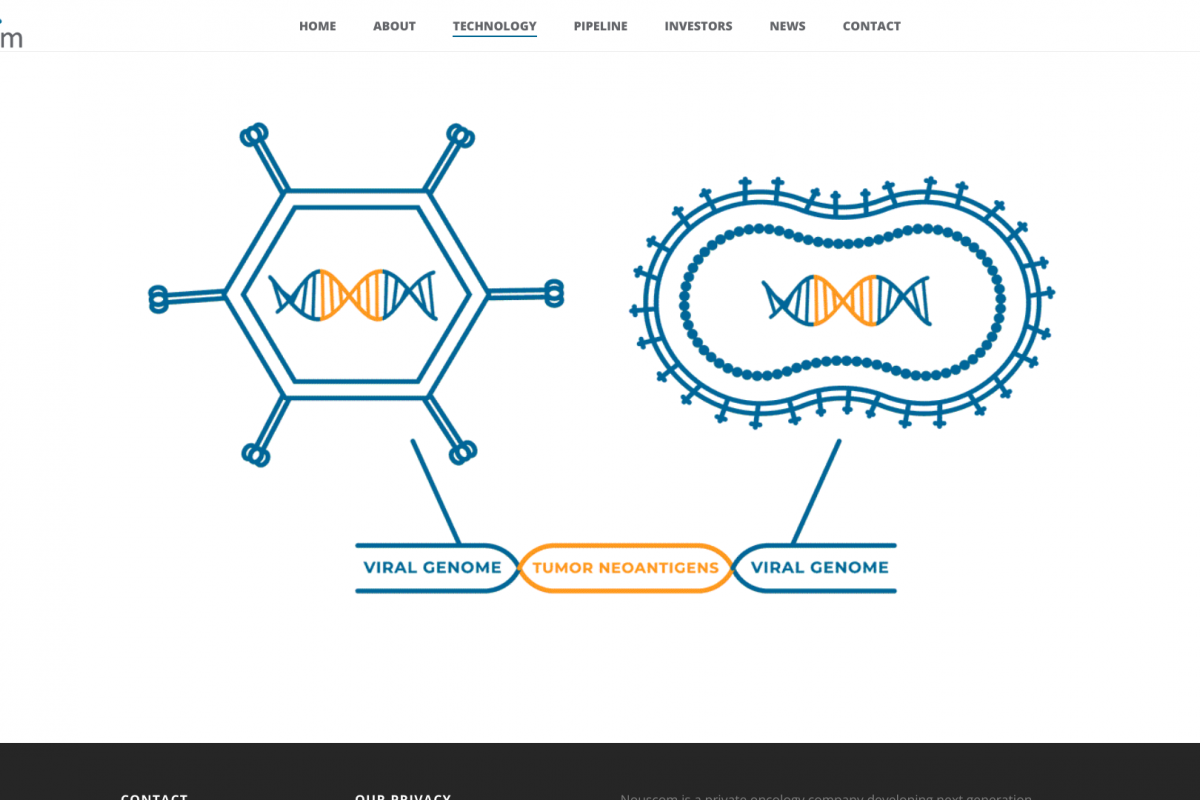
During the recent Respiratory Syncytial Virus (RSV) season, two types of immunization were available to protect young children from this respiratory disease.
As of March 20, 2024, the RSVVaxView Dashboard indicated about 60% of children were protected during the 2023-2024 season.
RSVVaxView reported that 43% of mothers with infants under eight months reported that their child had received Beyfortus™ (Nirsevimab), a single-dose, extended half-life monoclonal antibody offering passive immunization to prevent lower respiratory tract infections caused by RSV.
In February 2024, 38.8% of pregnant women reported that they plan to get nirsevimab for their infant, while 43.7% of women who are trying to get pregnant also reported that they plan to get nirsevimab for their future infant.
In early March 2024, the U.S. CDC reported that nirsevimab was 90% effective at preventing RSV-associated hospitalization in infants during their first RSV season.
With the increasing availability of nirsevimab in future RSV seasons, the CDC will assess its effectiveness over an entire season.
Additionally, among pregnant women with a gestational age of ≥32 weeks, the overall RSV vaccine coverage was about 17.9%.
As of January 31, 2024, the vaccination coverage was highest among non-Hispanic Asian (25.2%) pregnant women.
The CDC wrote, 'These RSV prevention products remain our single most important tool to protect infants during RSV outbreaks.'