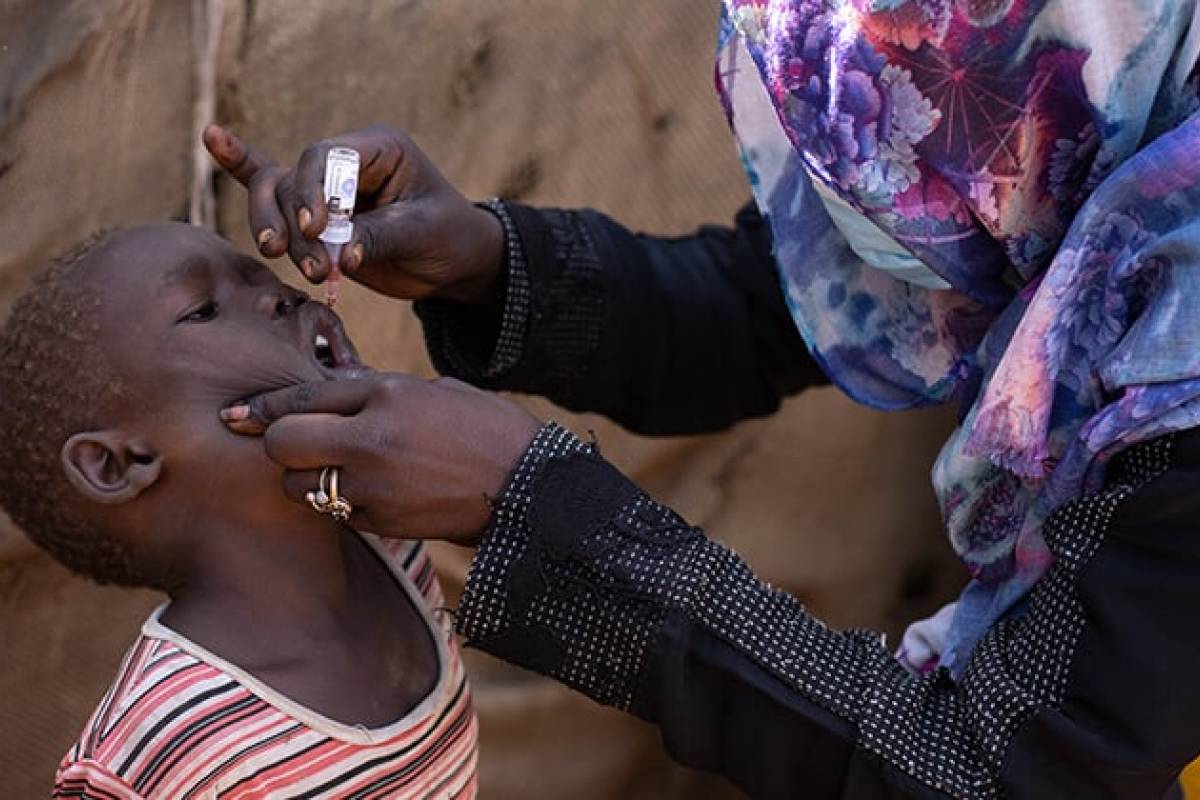
Following detecting a new emergence of variant poliovirus type 2 in six wastewater samples in the Port Sudan locality, Red Sea State, the Republic of Sudan recently announced polio vaccinations will begin in April 2024.
Sudan's Federal Ministry of Health (FMOH) announced on March 11, 2024, that this new detection comes 14 months after Sudan declared an outbreak of variant poliovirus type 2 from an emergence of the virus.
While no vaccination campaign has occurred since April 2023, surveillance for poliovirus in children focused on the most common indicator of a polio infection, acute flaccid paralysis.
While no child has been paralyzed due to the new emergence, detecting poliovirus in wastewater samples puts children across the country at high risk.
"The new detection has only redoubled our commitment to safeguarding our children's future. In collaboration with partners, we are mobilizing an outbreak response campaign to ensure that every child under five years in inaccessible areas receives the polio vaccine, and special plans will follow for hard-to-reach areas," said Dr. Dalya Eltayeb, Director-General of Primary Health Care in Sudan's FMOH, in a media statement.
In October 2022, the FMOH distributed 10.3 million doses of oral polio vaccine (OPV) in Sudan.
This year, Sudan is deploying the WHO-authorized type 2 novel oral polio (nOPV2) vaccine, which has been used 1 billion times by over 30 countries.
nOPV2 is reported to be more genetically stable than OPVs, with a lower risk of reversion to neurovirulence and less likely to mutate and cause paralysis.
While the nOPV2 vaccine is not approved in the United States, the government's vaccine committee considered its use under specific conditions during its February 28, 2024 meeting.