The outbreak of Marburg Virus Disease in Equatorial Guinea ended today with no new cases reported over the past 42 days after the last patient was discharged from treatment.
The outbreak, declared on February 13, was the first in Equatorial Guinea.
A total of 17 laboratory-confirmed cases and 12 deaths were recorded.
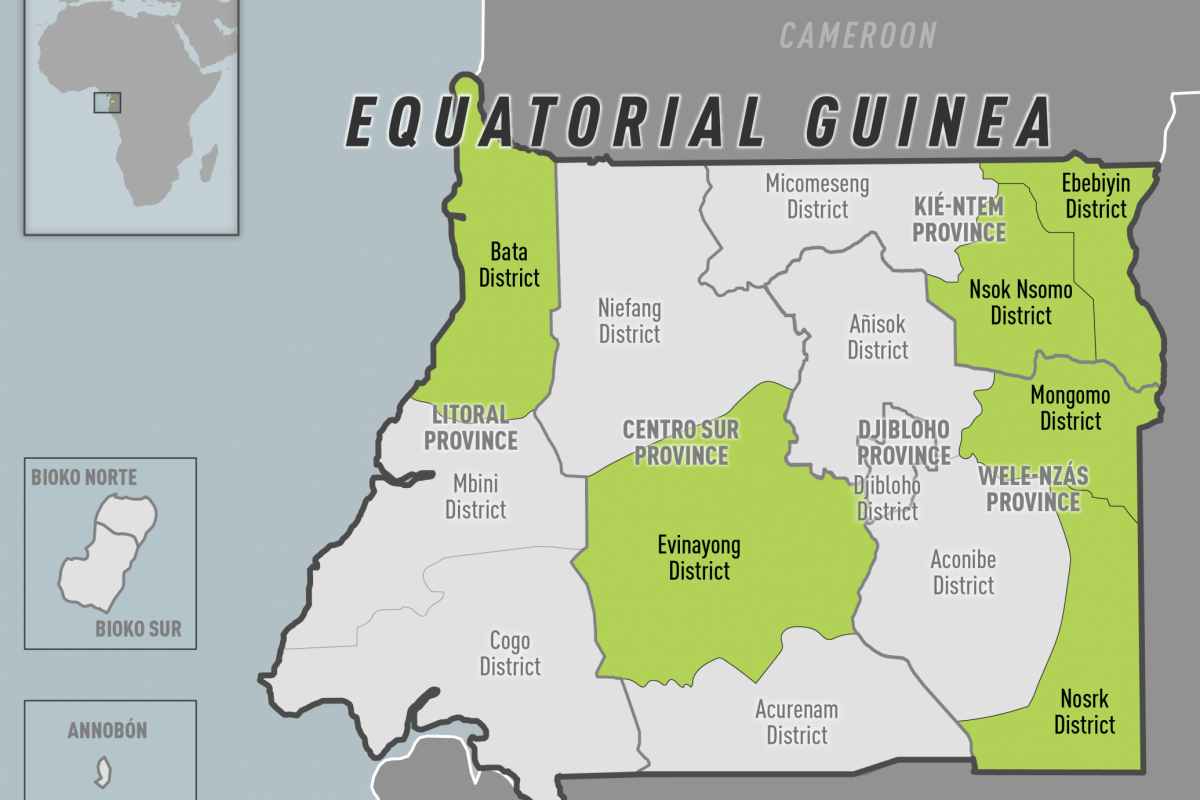
Five districts in four of Equatorial Guinea’s eight provinces were affected.
The western Litoral province Bata district was worst-hit, with 11 laboratory-confirmed cases reported. Among the reported cases, many were closely linked either through social gatherings and networks or geographically.
“While outbreak-prone diseases continue to pose a major health threat in Africa, we can bank on the region’s growing expertise in health emergency response to act quickly and decisively to safeguard the health and avert widespread loss of life,” said Dr. Matshidiso Moeti, World Health Organization (WHO) Regional Director for Africa, in a press release on June 8, 2023.
“The hard work by Equatorial Guinea’s health workers and support by partner organizations has been crucial in ending this outbreak. WHO continues to work with countries to improve measures to detect and respond effectively to disease outbreaks,” Dr Moeti added.
To support Equatorial Guinea’s response to the just-ended outbreak, WHO deployed experts in epidemiology, clinical management, health operations, logistics, risk communications, and infection prevention and control.
The Organization worked with the health authorities to set up a treatment center, provided medical supplies, including antivirals, and trained health workers in the critical aspects of outbreak control.
The WHO also supported the efforts by the authorities in neighboring Cameroon and Gabon to ramp up outbreak readiness and response.
Although the outbreak has ended, WHO continues to work with Equatorial Guinea to maintain measures such as surveillance and testing to enable prompt action should flare-ups of the virus occur, with the training provided during the outbreak helping to strengthen readiness capacity.
Marburg is in the same family as the virus that causes Ebola Virus Disease.
The Marburg virus is transmitted to people from fruit bats and spreads among humans through direct contact with the bodily fluids of infected people, surfaces, and materials.
In Africa, the first outbreak of Marburg was recorded in South Africa in 1975, followed by two others in Kenya in the 1980s. Since then, outbreaks have been reported in Angola, the Democratic Republic of the Congo, Ghana, Guinea, Uganda, and most recently, Equatorial Guinea and Tanzania.