Breaking News
RSV Therapy Gains Positive Vote to Protect Infants
June 8, 2023 • 2:58 pm CDT

by Jess Foami
(Vax-Before-Travel News)
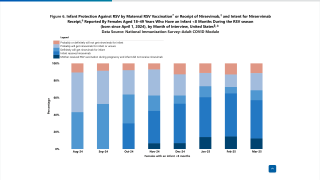
CNBC reported today the U.S. FDA's Antimicrobial Drugs Advisory Committee voted 21-0 to recommend full approval for Beyfortus® (Nirsevimab) for infants and young children.
The FDA considers committee recommendations when finalizing its decisions.
Beyfortu has already been approved in Canada, Europe, and the United Kingdom.
Beyfortus is the first extended half-life monoclonal antibody offering passive immunization to prevent lower respiratory tract infections caused by the respiratory syncytial virus (RSV).
AstraZeneca and Sanofi developed this RSV therapy.
This antibody therapy is not an RSV vaccine. The FDA recently approved two RSV vaccines for seniors.
Our Trust Standards: Medical Advisory Committee