Europe Authorized RSV Vaccine for Seniors

GSK plc today announced that the European Commission had authorized Arexvy™ for active immunization to prevent lower respiratory tract disease (LRTD) caused by a respiratory syncytial virus (RSV) in adults 60 and older.
The first launches are planned for the 2023/2024 RSV season, which typically starts in the autumn.
Tony Wood, Chief Scientific Officer, GSK, commented in a press release on June 7, 2023, "This authorization for Arexvy means eligible adults can be vaccinated against RSV disease for the first time, reinforcing GSK's long history of vaccine innovation."
Our strong manufacturing capability and scale, including from our vaccine manufacturing site in Belgium, means we are ready to deliver the (RSV) vaccine as countries begin to launch."
RSV is a common, contagious respiratory virus that leads to over 270,000 hospitalizations and approximately 20,000 in-hospital deaths each year in adults aged 60 years and over in Europe.
Those with underlying medical conditions, such as diabetes and chronic heart and lung disease, drive the majority of RSV hospitalizations.
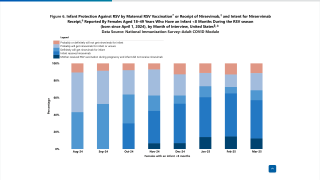
In the U.S., two RSV vaccines have been authorized for seniors, and one RSV monoclonal antibody (Beyfortus®) is seeking authorization for infants/children.
Our Trust Standards: Medical Advisory Committee