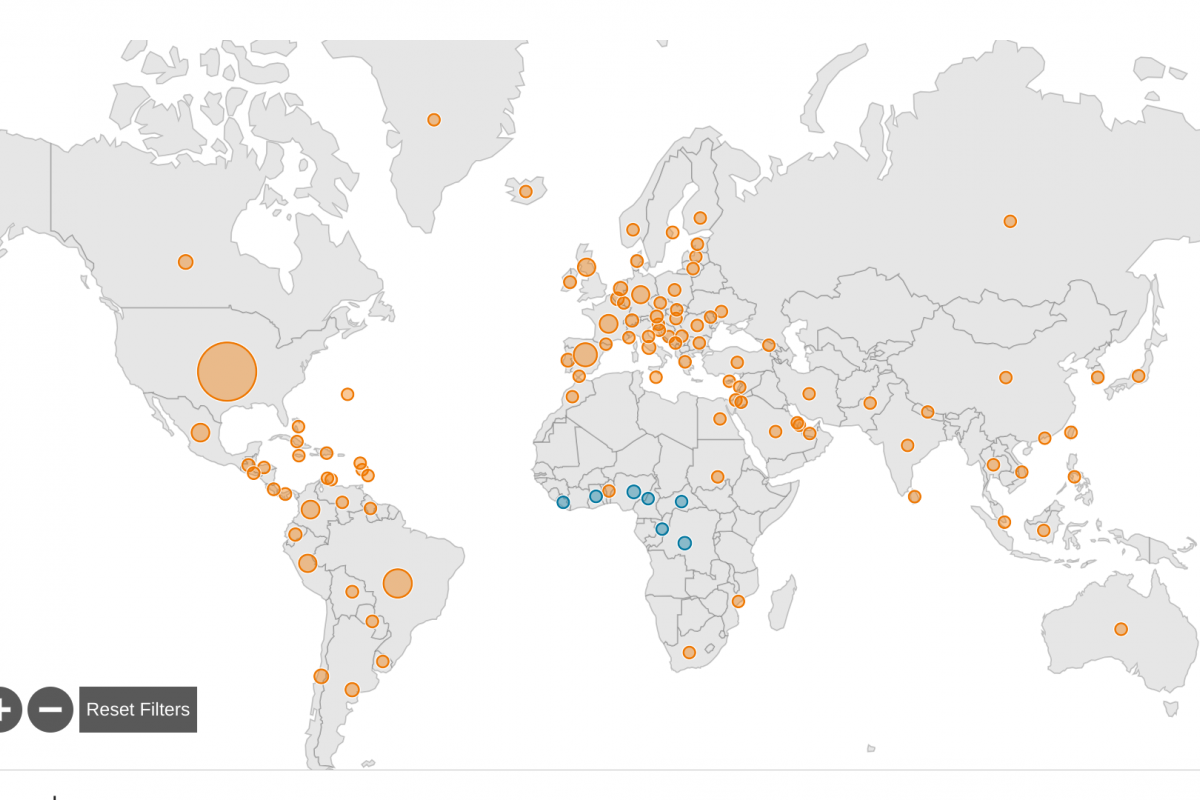
Since the last World Health Organization (WHO) situation report published in late June 2023, about seventeen countries have reported 316 new mpox cases and two new deaths have been reported by the WHO.
As of July 14, 2023, the WHO says there is a significant increase in mpox cases in the South-East Asia Region, driven by sustained community transmission in Thailand.
Available information on these Thailand cases shows that all patients are male, most from Bangkok without a travel history abroad in the 21 days before symptom onset. Around half of the new cases are among people living with HIV.
The U.S. CDC says the association of mpox cases with HIV infection highlights the need for a syndemic approach to care for HIV, sexually transmitted infections, and mpox in the context of comprehensive sexual health care.
However, mpox virus transmission continues at a low level in most countries reporting cases, and the main epidemiological and clinical characteristics of patients have remained stable over time, says the WHO.
From January 2022 through July 11, 2023, a cumulative total of 88,288 laboratory-confirmed cases of mpox, including 149 deaths, have been reported to WHO from 112 countries/territories/areas in all six WHO Regions.
And in the U.S., 1,243,378 mpox vaccine doses have been administered in 57 U.S. Jurisdictions.
As of July 15, 2023, Sexually Transmitted Disease vaccine news is posted by Precision Vaccinations.