RSV Vaccine Authorized in Great Britain

GSK plc recently announced that the Medicines and Healthcare products Regulatory Agency (MHRA) has authorized Arexvy for active immunization for the prevention of lower respiratory tract disease (LRTD) caused by respiratory syncytial virus (RSV) in adults 60 years of age and older.
This is the first time an RSV vaccine for older adults has been authorized in Great Britain.
Neale Belson, GSK's Senior Vice President and General Manager UK, said in a press release on July 10, 2023, "Our ambition is to help protect adults 60 years of age and older in the U.K. who are at risk from RSV disease, including those with underlying medical conditions, who drive the majority of RSV hospitalizations."
"This authorization for Arexvy means eligible adults can be vaccinated against RSV disease for the first time, reinforcing GSK's long history of vaccine innovation."
RSV is a common, contagious respiratory virus that leads to an estimated 175,000 GP visits, 14,000 hospitalizations, and 8,000 deaths yearly in adults aged 60 and over in the U.K.
Recent studies indicate that the burden of RSV disease may be even greater than that of influenza in hospitalized older adults.
In the U.S., two RSV vaccines have been approved, and several RSV vaccine candidates are conducting late-stage clinical trials.
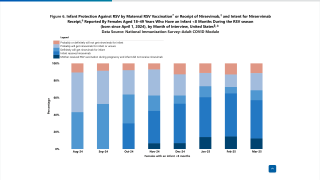
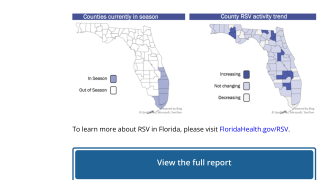
Precision Vaccinations publishes RSV seasonal trends for 2023.
Our Trust Standards: Medical Advisory Committee