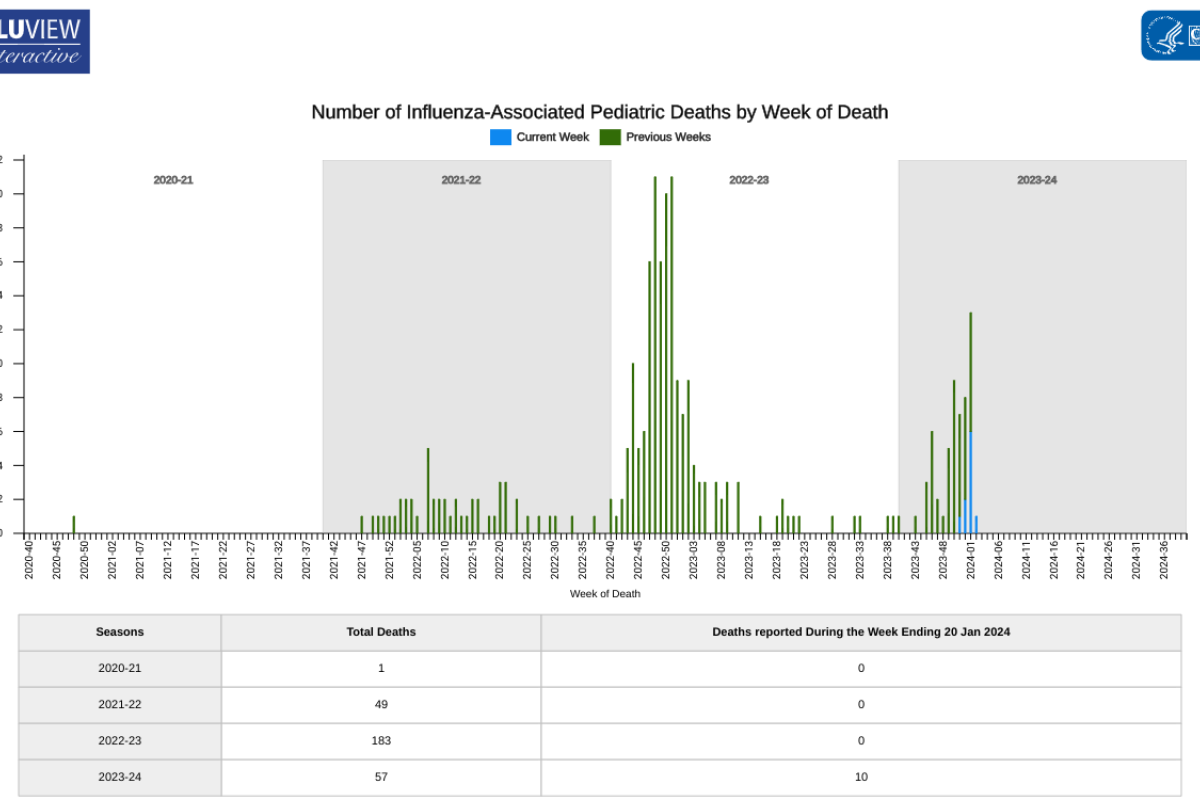
According to new data posted by the U.S. Centers for Disease Control and Prevention (CDC), ten additional children died from influenza last week.
Six deaths were associated with influenza A viruses, such as A(H1N1).
Three deaths were associated with influenza B viruses with no lineage determined.
One pediatric death was associated with a co-infection with influenza A(H1N1) and influenza B/Victoria viruses.
As of January 26, 2024, the CDC's FluView dashboard indicates a total of 57 influenza-associated pediatric deaths have now occurred during the 2023-2024 flu season.
During the 2022-2023 flu season, 183 children died from influenza infections. The CDC has not indicated the vaccination status of the fatalities.
From an overall impact perspective, the National Center for Health Statistics reported from October 2, 2022, to September 9, 2023, of all deaths in the U.S., 9,697 (4%) of death certificates listed influenza.
While the CDC stated last Friday that key flu indicators have decreased or remained stable nationally for three weeks, there is an ample supply of flu shots (egg, cell, and nasal-based) offered at most pharmacies in the U.S.
Over 156 million flu vaccines had been distributed during the 2023-2024 season. This decreased from the 173 million influenza vaccines distributed last flu season.