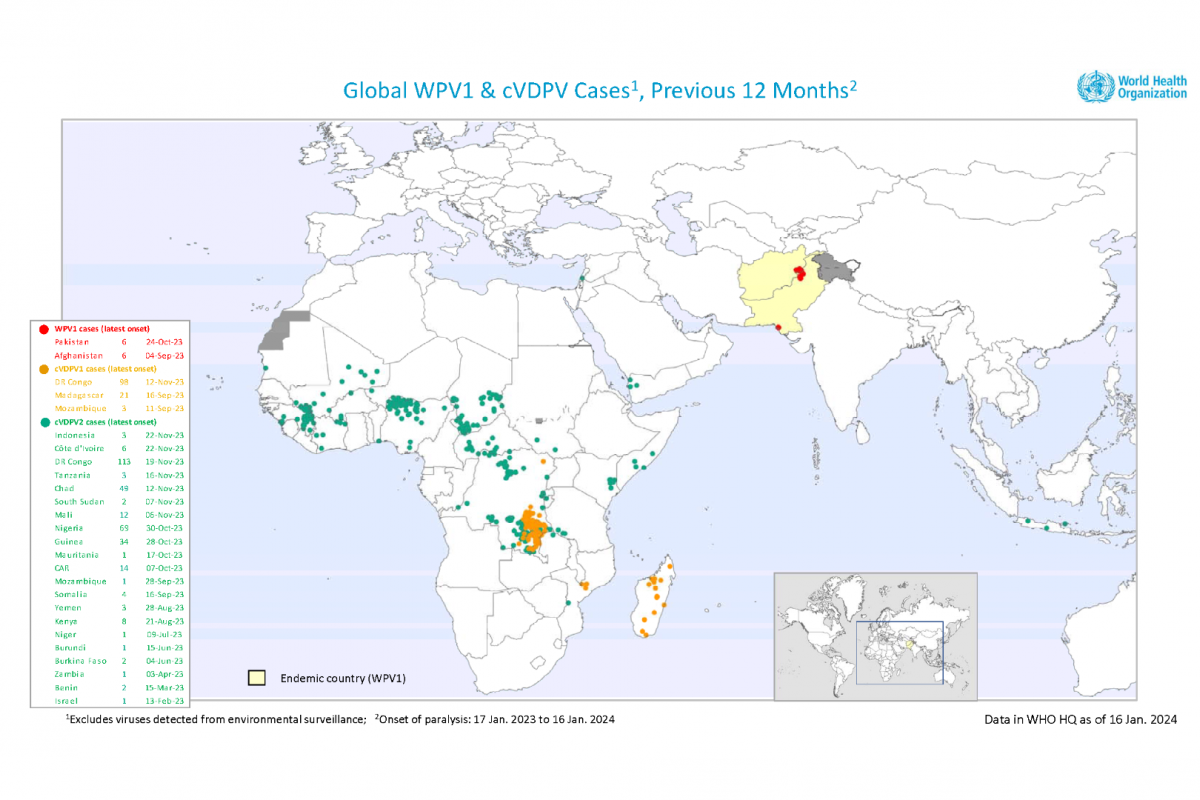
The Global Polio Eradication Initiative (GPEI) recently reported 20 circulating vaccine-derived poliovirus type 1 or type 2 (cVDPV2) cases were reported in six African countries in late 2023.
Chad, the Democratic Republic of the Congo, Ivory Coast, Mozambique, Nigeria, and South Sudan reported new polio cases.
Since March 2021, the GPEI has helped administer nearly 1 billion doses of novel oral polio vaccine type 2 (nOPV2) across 35 countries, primarily in Africa.
After nearly three years of use, estimates show that nOPV2 is 80% less likely to seed new variant polio outbreaks, making it the tool of choice to stop these outbreaks for good, says the GPEI.
The World Health Organization (WHO) confirmed in December 2023 that the spread of the poliovirus (31 countries) remained a Public Health Emergency of International Concern.
The WHO Director-General, Dr Tedros Adhanom Ghebreyesus, commented on January 9, 2024, "nOPV2 has blazed a trail for other new vaccines that address critical health emergencies."
In late December 2023, the WHO issued its first-ever prequalification approval for a vaccine, nOPV2, being used under its Emergency Use Listing regulatory pathway.
As of early January 2024, 325 cases of cVDPV2 had been reported in 2023, compared to 689 cases in 2022.
In the United States, following the detection of poliovirus in New York, the U.S. CDC selected jurisdictions to strategically expand wastewater testing for poliovirus in counties with potentially low polio vaccination coverage.