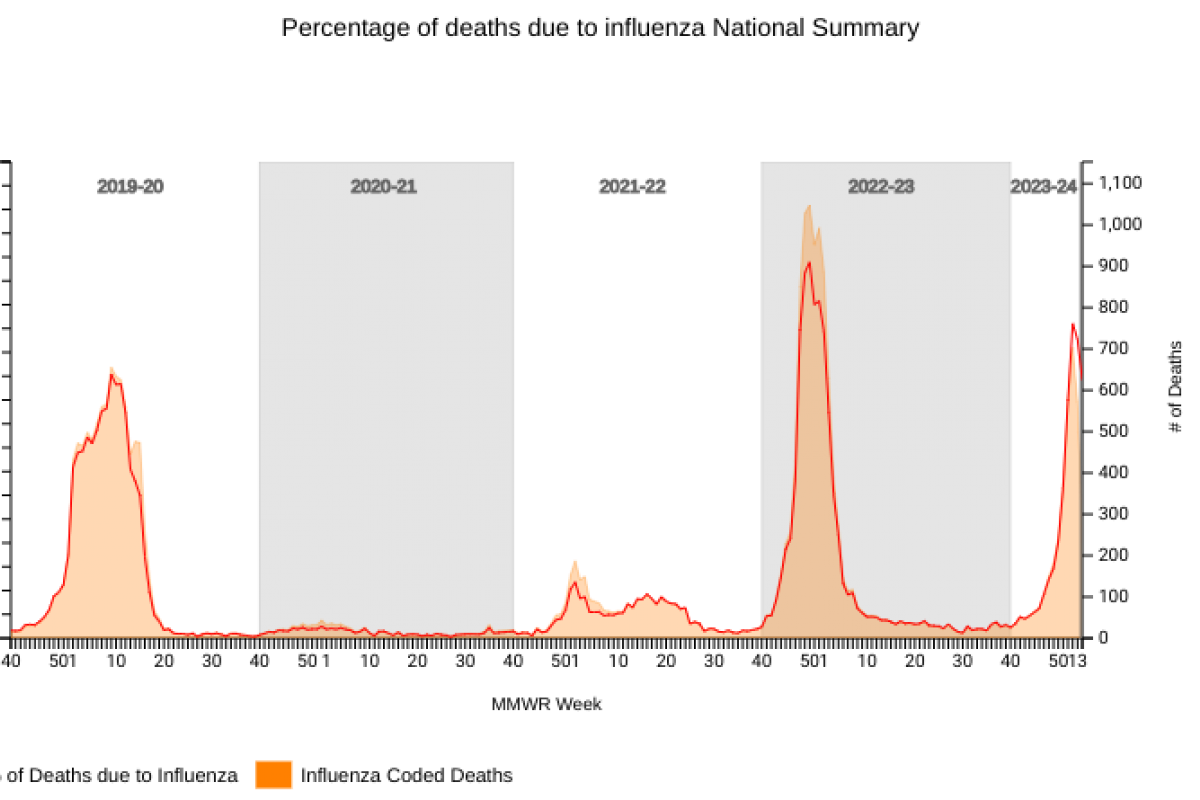
According to new data posted by the National Center for Health Statistics (NCHS) Mortality Surveillance, 0.9% of all deaths in the U.S. that occurred during the week ending January 27, 2024 (Week 4), were due to influenza.
On February 2, 2024, the U.S. CDC stated this percentage decreased by ≥ 0.1 compared to Week 3.
During week 1, with about 100% of the data tabulated, there were 703 influenza-related deaths reported by the NCHS.
From October 2, 2022, to September 9, 2023, 9,697 (4%) deaths were listed as influenza.
Additionally, the CDC reported eight influenza-associated pediatric deaths during the 2023-2024 season, which were reported to the CDC during Week 4. A total of 65 influenza-associated pediatric deaths have occurred this flu season and have been reported by the CDC.
Last flu season, there were 178 influenza-associated pediatric fatalities. The mean age at death was about seven years, and 83% of these children were not fully vaccinated.
The CDC continues to recommend annual flu shots (egg, cell, or nasal-based) for most people and booster vaccinations for some people after conferring with a healthcare provider.