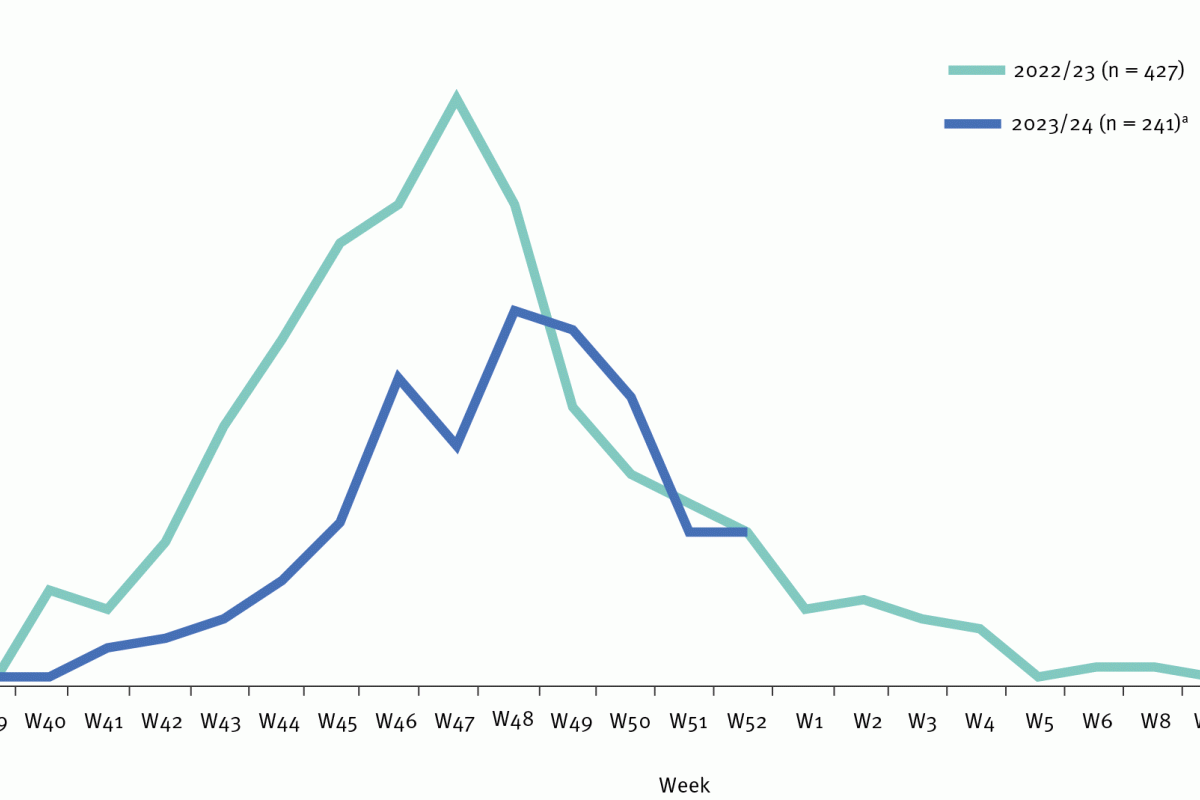
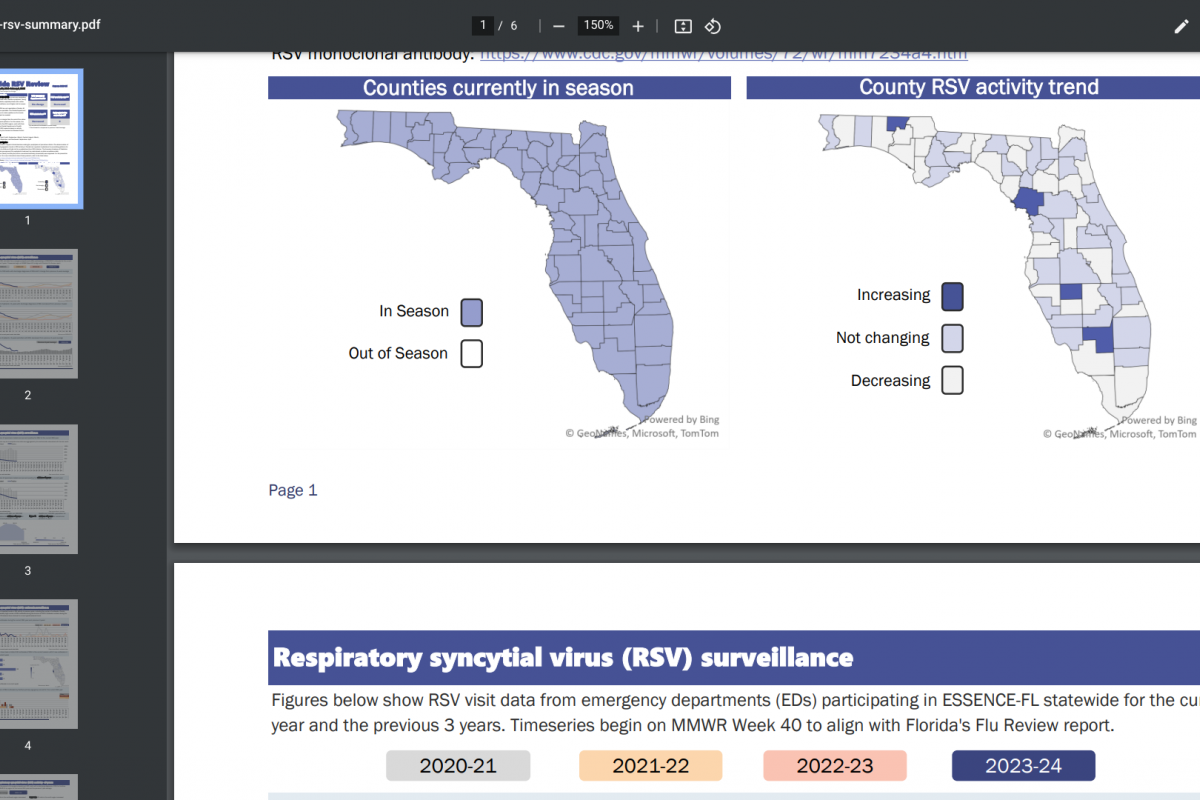
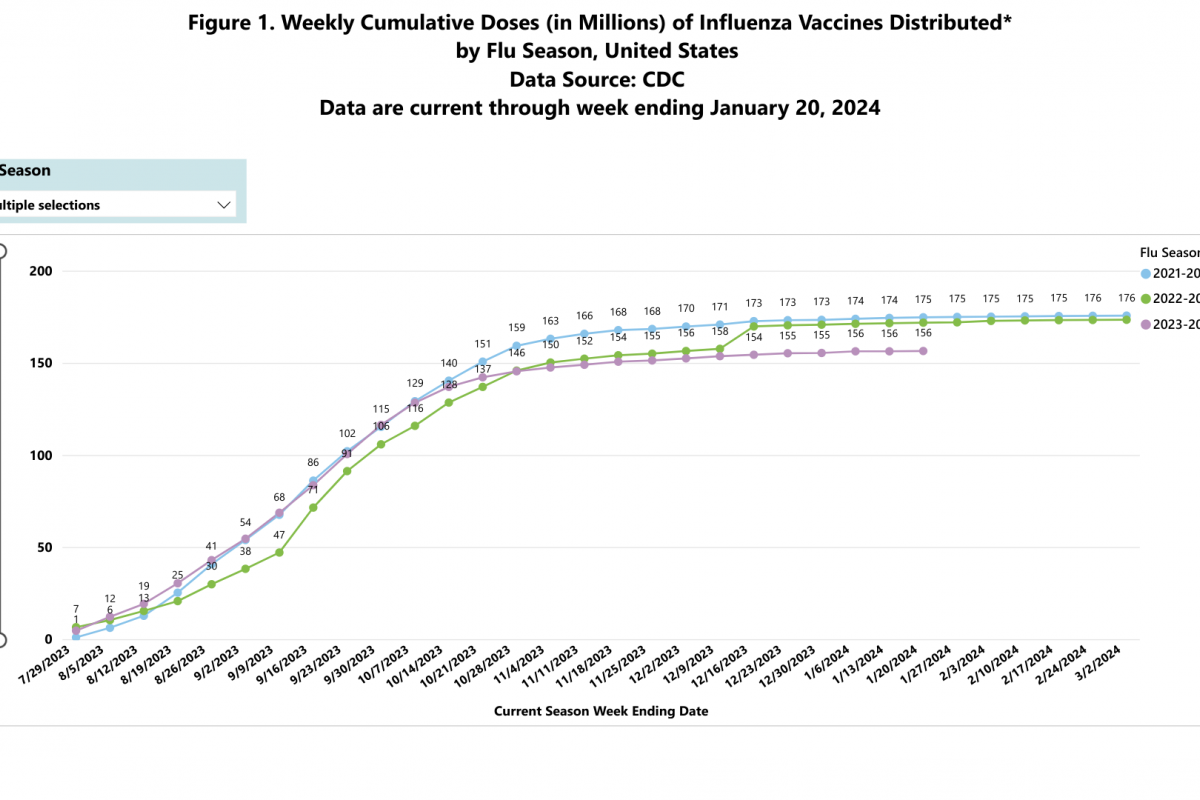
Throughout the 2023-2024 respiratory syncytial virus (RSV) season, newly approved vaccines have been offered to pregnant women and older adults. As with all vaccines, it takes time to appreciate their ability to protect people from disease fully.
According to a report by TD Cowen's analyst Tyler Van Buren on February 8. 2024, Moderna Inc.'s mRNA-based RSV vaccine candidate may not be as effective as its competitors.
The Wall Street firm's report cites a Phase 3 clinical trial, which found that Moderna's mRNA-1345 vaccine candidate has an overall efficacy of 63.3% against two-symptom RSV disease after a follow-up of 8.6 months.
This is a significant change from a January 2023 reading, which showed mRNA-1345 had an efficacy of 84%.
"In the absence of head-to-head clinical trials, comparative conclusions regarding the safety and efficacy of mRNA-1345 relative to other RSV vaccines cannot be made," Moderna said in this abstract.
Moderna has previously confirmed it has submitted regulatory filings to the FDA for its RSV vaccine, indicating potential approvals ahead of the 2024-2025 RSV season in the U.S.
As of February 9, 2024, the U.S. CDC estimated the percentage of adults 60+ receiving an RSV vaccine was 22.4%. As of January 27, 2024, certain pregnant women's overall RSV vaccination rate was 16.2%.