Doctors Without Borders/Médecins Sans Frontières (MSF) recently announced it is gravely alarmed over the depletion of the global oral cholera vaccine (OCV) stockpile.
As of March 6, 2024, all cholera vaccine doses currently in production have already been allocated, and their demand keeps increasing.
The predicted shortfall of at least 50 million vaccine doses worldwide.
As countries report cholera outbreaks, MSF says that existing and new manufacturers must urgently step up their efforts to boost the global supply of cholera vaccines.
MSF says immediate action is necessary to address this pressing global health concern.
"The world's oral cholera vaccine stocks have dried up," said Dr. Daniela Garone, MSF's international medical coordinator, on February 26, 2024.
"We're very worried that people are at much greater risk of getting and potentially dying from a disease that's preventable if there's no vaccine available to respond to an outbreak in their community, nor to prevent one from starting in the first place," Dr. Garone added.
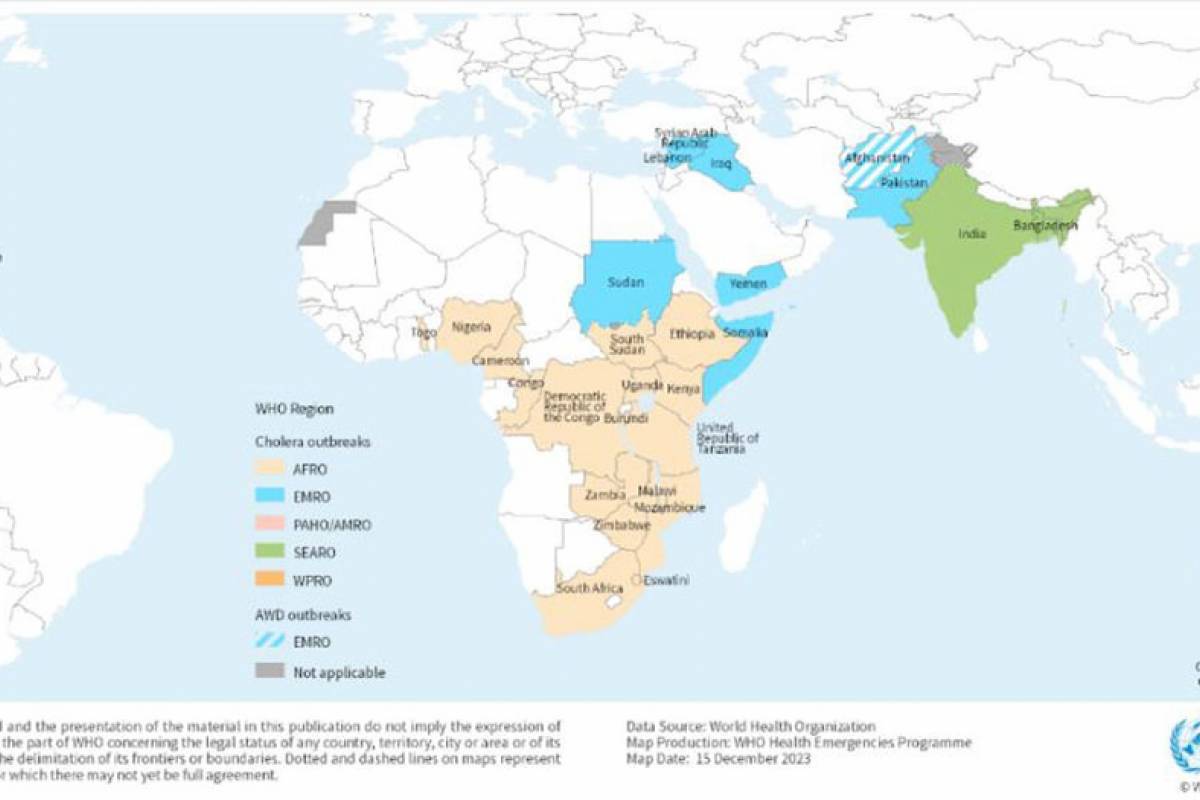
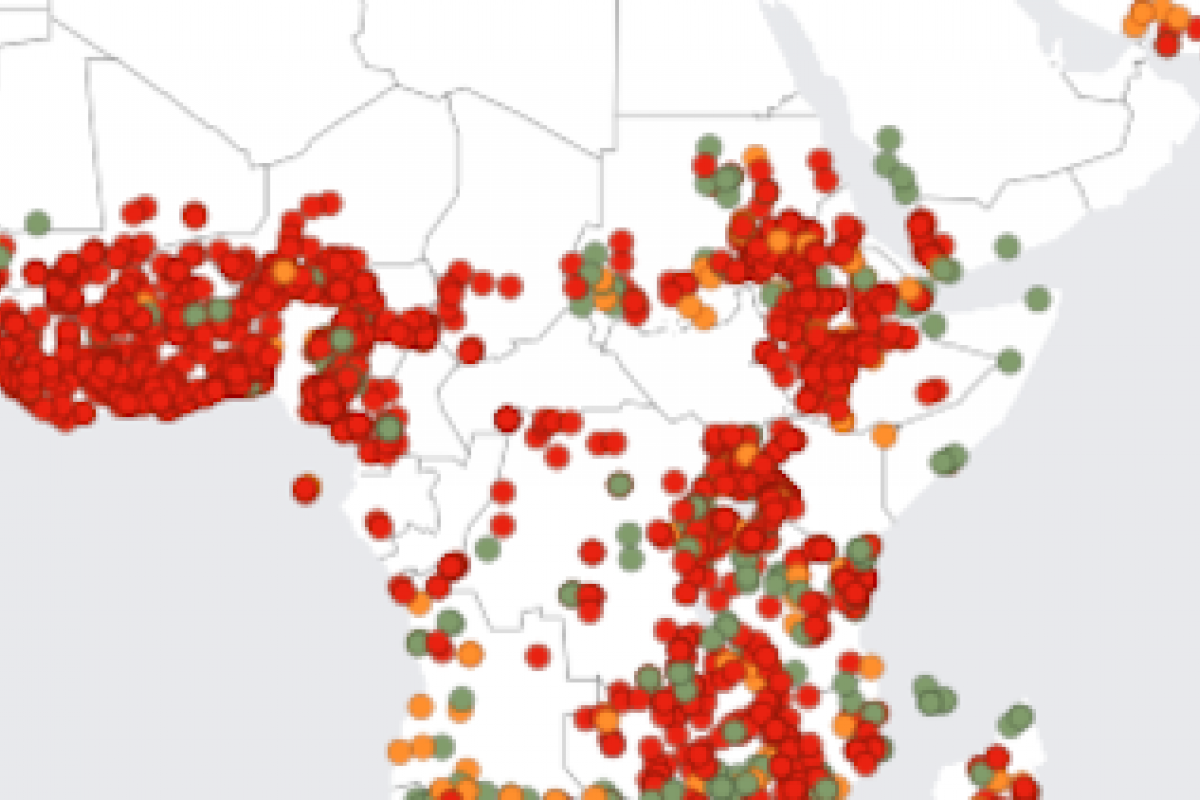
The World Health Organization (WHO) has recorded seven cholera pandemics over the past two centuries. The current (7th) cholera epidemic is considered to have started in 1961.
About 17 countries across four WHO regions reported over 40,000 cholera cases and 775 related deaths in January 2024.
The five countries reporting the most cases in 2024 are Zambia, Afghanistan, Zimbabwe, Mozambique, and Pakistan.
The U.S. CDC recommends that adults traveling to areas with active cholera transmission get vaccinated.
The WHO has pre-qualified three vaccines to address this need: Dukoral®, Shanchol™, and Euvichol®.
DUKORAL is authorized in Australia, Canada, Europe, New Zealand, Thailand, and the United Kingdom to protect people against cholera and Enterotoxigenic Escherichia coli.
DUKORAL vaccine sales were reported to be €29.8 million in 2023 compared to €17.3 million in 2022, a 72% increase.