India Launches Free HPV Shots For Young Women With Older Vaccine

Cervical cancer continues to pose a significant public health challenge worldwide, including in India, where it results in over 42,000 deaths each year.
In response to this pressing health issue for women, India's Union Health Ministry is launching a free nationwide vaccination program against the Human Papillomavirus (HPV) to help prevent this largely vaccine-preventable disease.
Set to begin in late February 2026, this initiative aligns with global efforts led by the World Health Organization (WHO) to eliminate cervical cancer by 2030. In India, approximately 12 million girls will be eligible for vaccination each year.
Following WHO recommendations, the program will provide a single-dose regimen for adolescents, which has demonstrated strong immunity with efficacy rates exceeding 93% against persistent HPV infections. An optional second dose may be offered after 3 to 5 years if needed.
The vaccine supply is supported by Gavi, the Vaccine Alliance, which has secured millions of doses to ensure access across the Indian subcontinent. Vaccinations will be administered voluntarily and at no cost through government health facilities, including Ayushman Arogya Mandirs, community health centers, district hospitals, and medical colleges.
For this national program, India has selected Gardasil. This quadrivalent recombinant vaccine protects against four key HPV types: high-risk oncogenic strains 16 and 18, responsible for approximately 70% of cervical cancers globally, as well as low-risk types 6 and 11, which cause about 90% of genital warts.
Meanwhile, the locally produced Cervavac vaccine is still commercially available in India's private sector. Approved in 2022, it is India's first domestically produced quadrivalent HPV vaccine.
While India's selection of the older Gardasil vaccine prioritizes cost-effectiveness and core protection against the most prevalent cancer-causing strains in the region, the U.S. offers a nonavalent option that provides broader coverage.
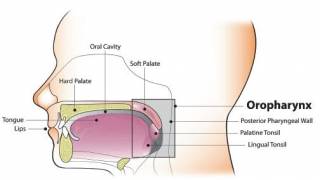
The leading HPV vaccine in the U.S. is Gardasil 9, recommended by the Centers for Disease Control and Prevention (CDC) for routine immunization starting at ages 11-12. Gardasil 9 expands protection to nine HPV types, preventing up to 90% of cervical cancers and additional HPV-related conditions such as anal, oropharyngeal, and vulvar cancers.
Notably, the U.S. CDC updated its guidelines in January 2026 to recommend a single-dose schedule for children, reflecting emerging evidence of long-term efficacy from just one dose.
The CDC recommends consulting healthcare providers for personalized advice on HPV vaccination.
Our Trust Standards: Medical Advisory Committee