$37 Million Funds Vaccine Candidates Targeting Gonorrhea and Shigellosis

LimmaTech Biologics AG announced today the closing of a USD 37 million Series A financing round that will empower its proprietary technology platform and accelerate its preclinical and clinical vaccine candidates against increasingly dangerous bacterial infections, including programs addressing shigellosis and gonorrhea.
Antimicrobial Resistance is responsible for approximately 5 million deaths annually. Infections that were once easily treatable have now become difficult, if not impossible, to cure.
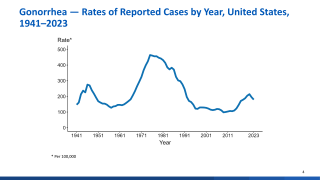
As a leading example of this threat to global health, half of the approximately 700,000 annual gonorrhea infections in the U.S. are already resistant to antibiotics, and there is a real threat of gonorrhea soon becoming untreatable.
While there are no gonorrhea vaccines available, off-label vaccines and treatments are in use.
Later-stage clinical development efforts will focus on the company's Shigella vaccine program, which LimmaTech developed with GSK. The company expects to announce preliminary results from the Shigella program's ongoing Phase 2 clinical trial in the second half of 2023.
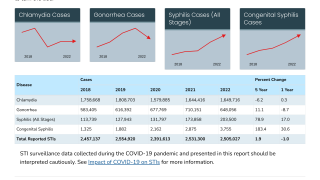
Shigella cause an estimated 450,000 infections in the U.S. each year.
According to the U.S. CDC, people can get a Shigella infection (shigellosis) after putting something in their mouth or swallowing something that has come into contact with the stool of someone with a Shigella infection.
"Within the next decade, multiple bacterial infections will become untreatable due to antimicrobial Resistance, which is already a significant burden on global health. By advancing our innovative technology platform, LimmaTech has the potential to simultaneously provide vaccine-induced protection against bacterial infections, mitigate the increasing risk of antibiotic resistance, and move toward the control of several highly transmissible pathogens," commented Dr. Franz-Werner Haas, CEO of LimmaTech, in a press release on October 9, 2023.
".....With this support and our team of proven experts in bacterial vaccine development and manufacturing, we look forward to providing life-changing vaccines to address a major global medical need."
The Company is conducting a Phase I/II clinical trial in the Republic of Kenya of a 4-valent candidate vaccine to help prevent diarrheal disease caused by the Shigella bacteria in children and infants in low and middle-income regions. The Shigella study is conducted in collaboration with GSK and the Wellcome Trust.
LimmaTech is committed to translating novel scientific concepts into highly effective vaccines that benefit humanity. For more information, please visit www.lmtbio.com.
Our Trust Standards: Medical Advisory Committee