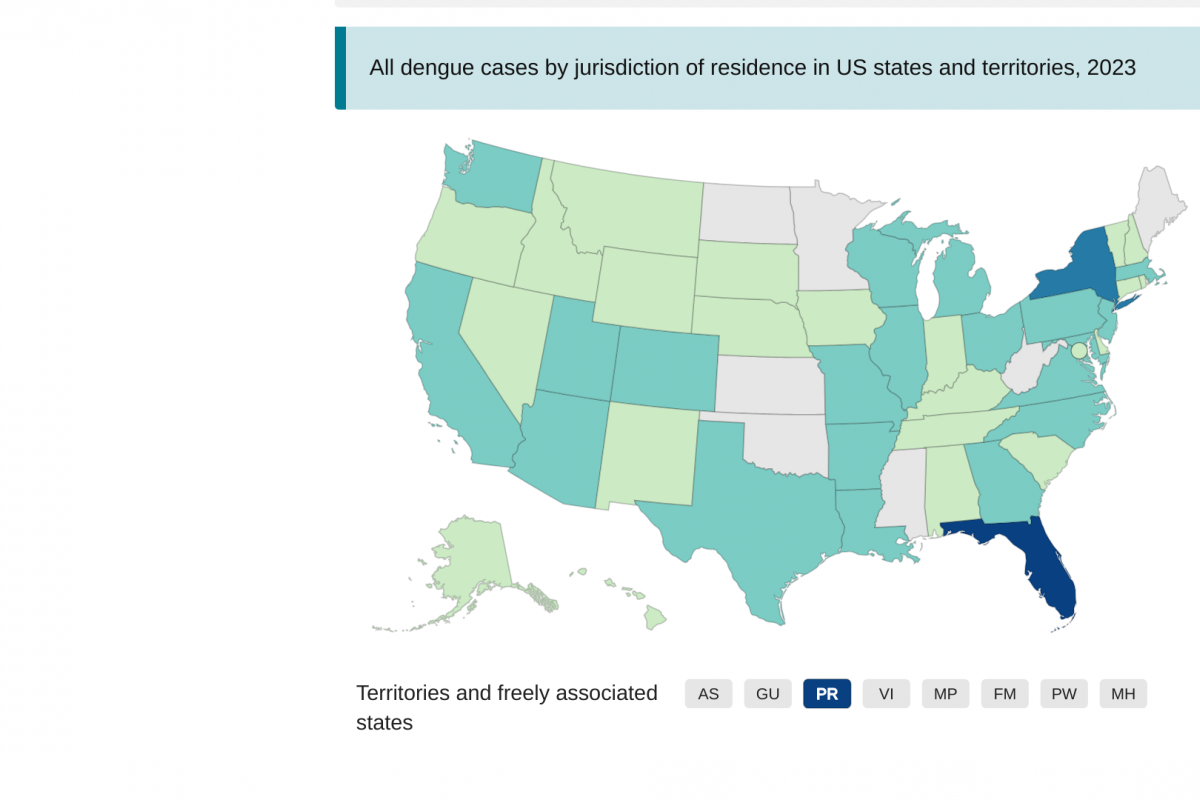
Throughout 2023, the Florida Health Department has been reporting travel-related and locally acquired dengue cases. Dengue is a vectorborne infectious disease spread by infected mosquitoes and endemic in about 125 countries.
During week #40, Florida reported 32 new dengue cases.
As of October 7, 2023, Florida's Arbovirus Surveillance report confirmed 17 new travel-associated Dengue cases. In 2023, there have been 351 dengue cases associated with international travelers, led by Miami-Dade County with 207.
Over sixty-two percent (220) of these dengue cases are related to visitors from Cuba.
Of more concern to health officials is the recent increase in locally acquired dengue cases.
Furthermore, there were 15 new cases of locally acquired dengue last week. This data increases 2023's total to 53 cases, with Miami-Dade County reporting 47 locally acquired cases.
According to the Centers for Disease Control and Prevention, there have been 1,289 dengue cases reported by 48 U.S. jurisdictions in 2023. In addition to Florida, New York has reported 90 dengue cases this year.
About 3.4 million dengue cases have been reported in the Region of the Americas so far this year.
As of October 11, 2023, two dengue vaccines are available globally, but only one is licensed in the U.S. Sanofi Pasteur's Dengvaxia® vaccine is available for certain people following a diagnostic test review.