The Global Polio Eradication Initiative (GPEI) recently confirmed six countries reported additional polio cases in 2023
These countries include Pakistan, which reported its fifth wild poliovirus type 1 (WPV1) case this year.
According to the GPEI, the other countries include Chad, the Democratic Republic of the Congo (DRC), Guinea, Nigeria, and Somalia, which reported more circulating vaccine-derived poliovirus type 2 cases.
The DRC also reported four more cases involving circulating vaccine-derived poliovirus type 1.
While it has been about three months since the World Health Organization (WHO) confirmed that the spread of the poliovirus remained a Public Health Emergency of International Concern, the WHO continues to remind Member States that the risk of the emergence of polio has increased due to low vaccination coverage.
Polio is a crippling and potentially deadly disease that affects the nervous system.
Most people with polio do not feel sick. In rare cases, polio infection causes permanent loss of muscle function (paralysis). Polio can be fatal if the muscles used for breathing are paralyzed or if there is an infection of the brain.
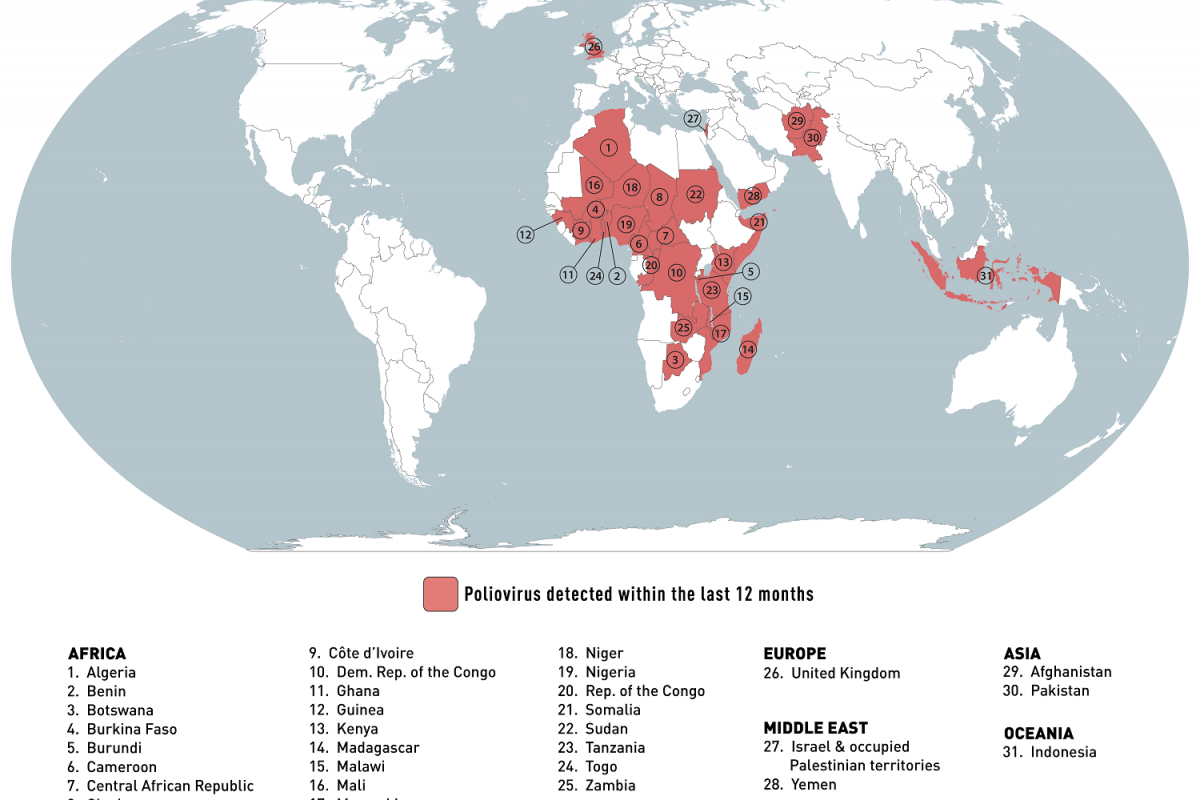
To warn international travelers, the U.S. Centers for Disease Control and Prevention issued an updated Global Polio Alert - Level 2, Practice Enhanced Precautions, Travel Health Notice on September 11, 2023, regarding polio outbreaks and poliovirus detections in 31 countries.
The CDC says that before visiting any of these destinations, adults who previously completed the full, routine polio vaccine series may receive a single, lifetime booster dose of the polio vaccine.
As of November 2023, various polio vaccines are available worldwide.