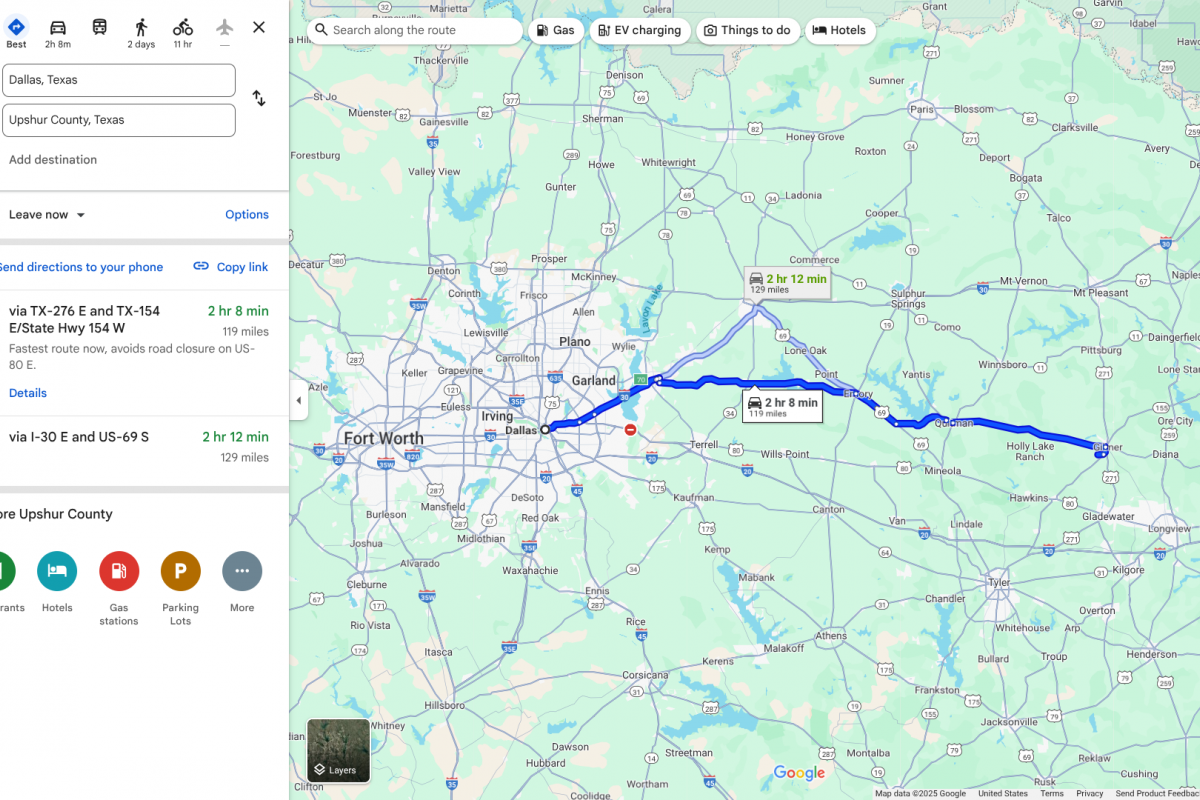
The epicenter of the measles outbreak in Texas appears to have moved about 120 miles east of Dallas to the sparsely populated Upshur County.
According to the Texas Department of State Health Services (DSHS) and a health department Facebook post, about 19 confirmed cases of measles were reported at a single location in Upshur County.
All cases involve individuals over the age of 17, with unknown vaccination status, who were linked to two individuals who visited Upshur County from outside the state. As of April 19, 2025, all affected persons were isolated from the public and are following all the appropriate guidelines.
These measles cases have not included any Upshur County residents, and there are no reports of measles in any of our Upshur County public schools, public buildings, county operations, or medical facilities.
DSHS stated that it is gathering additional information to determine the residency status of these cases and whether they are related to the West Texas measles outbreak in 2025, which has reached 597 cases.
Other counties in Texas have reported 10 measles cases in 2025.
Throughout the United States, 25 jurisdictions have reported about 800 measles cases this year.