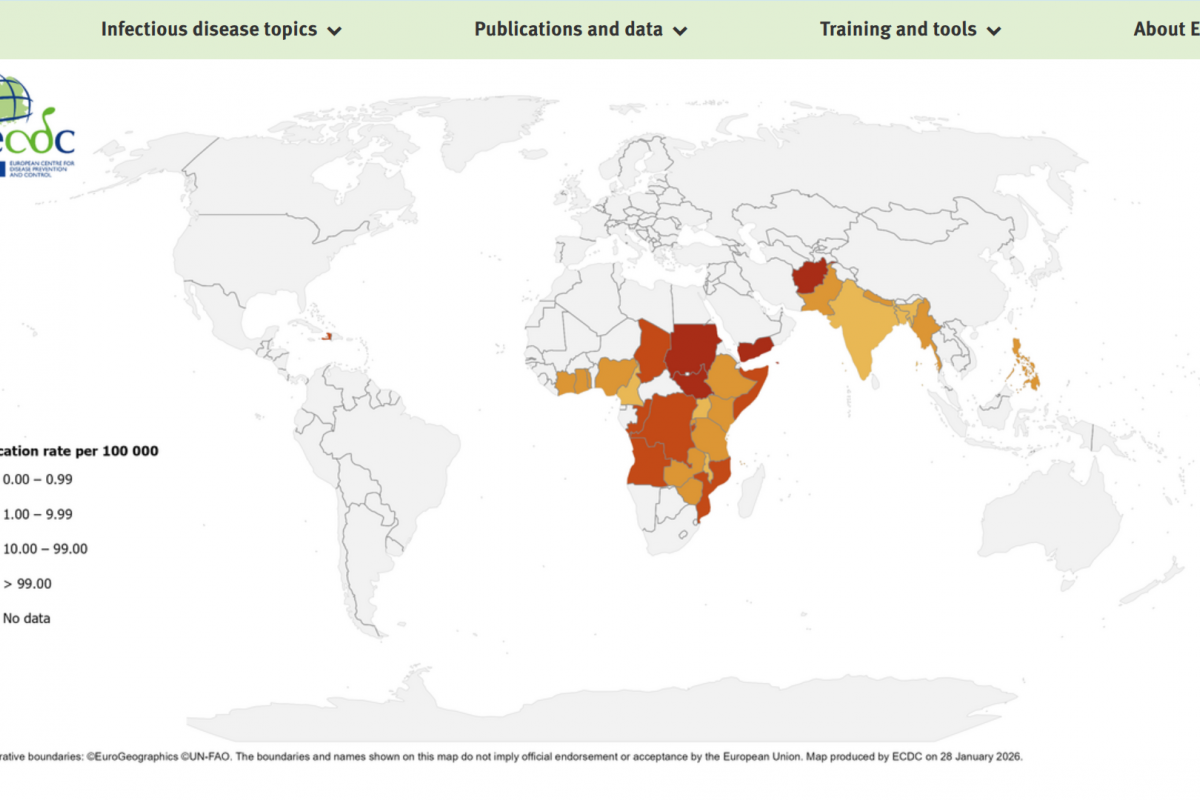
The early weeks of 2026 have seen a significant decline in reported cholera cases compared to the same period last year, according to preliminary data from the European Centre for Disease Prevention and Control (ECDC) in its week #5 report.
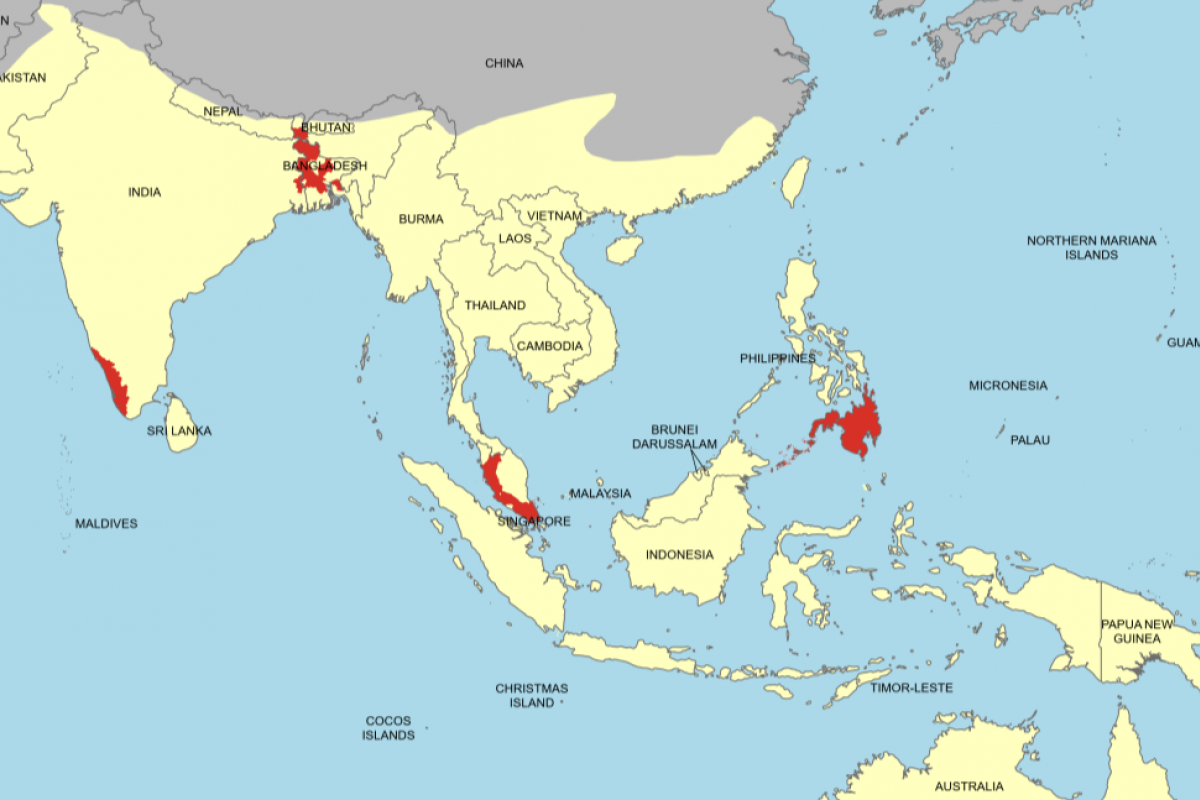
The ECDC reported that from January 1 to January 27, 2026, a total of 1,203 cholera cases, including seven deaths, were reported across eight countries: Angola, Burundi, Ethiopia, Malawi, Mozambique, Namibia, Somalia, and Zambia.
Health authorities attribute the decline to a combination of seasonal factors, improved response efforts, and expanded interventions in high-risk areas.
This figure marks a significant decrease from the equivalent period in 2025, when 10,043 cases and 32 deaths were reported worldwide by January 27. The reduction aligns with broader trends observed in late 2025, when global cholera activity began to ease in some regions after peaks in previous years.
For context, the World Health Organization (WHO) reported a cumulative total of approximately 614,828 cholera cases and 7,598 deaths from January 1 to December 28, 2025, across 33 countries in five WHO regions.
Vaccine availability is a critical concern in controlling disease outbreaks.
The global oral cholera vaccine (OCV) stockpile, managed by the International Coordinating Group (ICG) and partners such as UNICEF and WHO, has fluctuated in recent years as high demand has outstripped production.
As of early 2026, emergency stockpile reports indicate that over 20 million doses are available (including formulations such as OCV-S), with ongoing production increases aimed at meeting global needs.
WHO-prequalified OCVs, such as Vaxchora, are being deployed for reactive vaccination campaigns in affected areas.
The ECDC and the WHO say that international travelers to affected regions should consult healthcare providers and local travel vaccination experts about OCV options to prevent disease while abroad.