The U.S. CDC's Advisory Committee on Immunization Practices (ACIP) workgroup focused on polio and poliovirus today proposed updated guidance regarding vaccinations in the U.S.
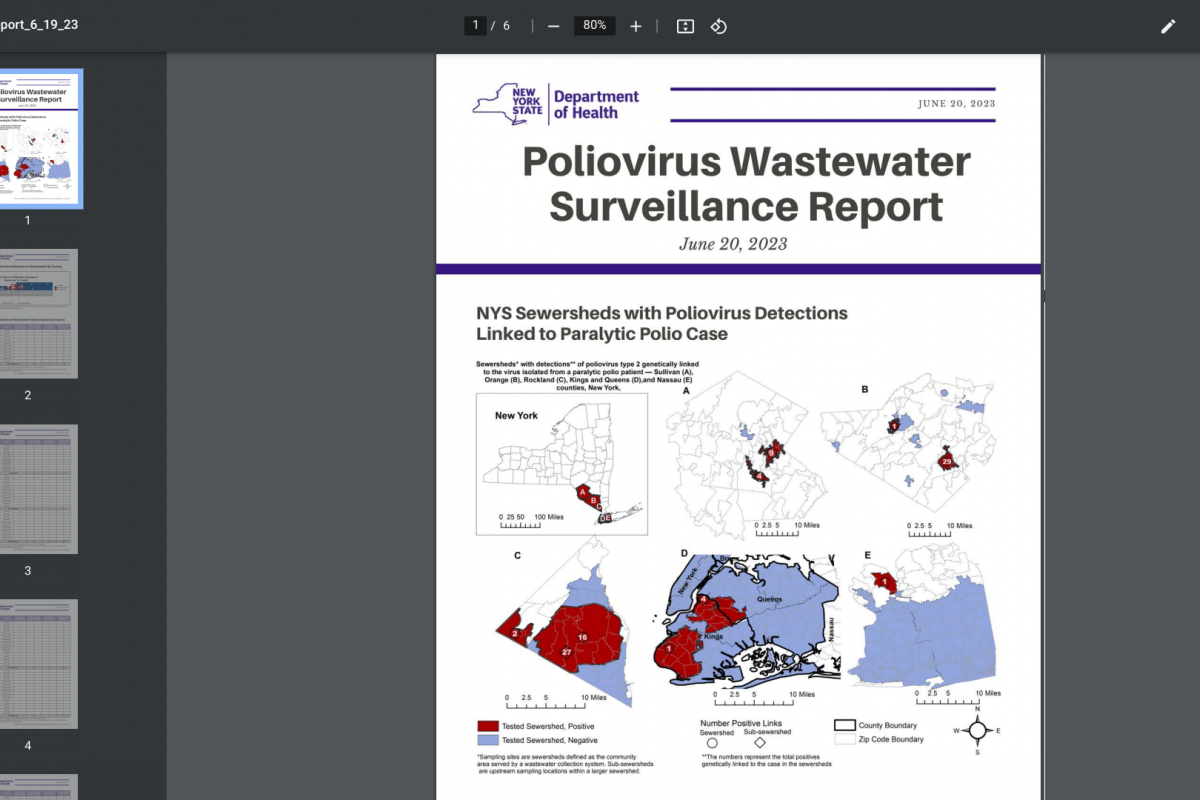
The ACIP began debate on updating its vaccination recommendation following one paralytic polio case in an unvaccinated adult in Rockland County, NY was confirmed in 2022.
Furthermore, wastewater testing confirmed poliovirus detections in several New York counties in 2022 and 2023.
The CDC stated this finding likely indicated about 1 to 2 thousand people in southern New York had mostly asymptomatic infections.
As of June 21, 2023, the ACIP committee is voting on this language: Adults who have received a primary series of tOPV or inactivated poliovirus vaccine (IPV) in any combination and are at increased risk of poliovirus exposure may receive another dose of IPV.
And the available data do not indicate the need for more than a single lifetime booster dose with IPV for adults.
Sanofi Pasteur's single-antigen Imovax Polio® (Ipol®) is a highly purified IPV with enhanced potency, says the U.S. FDA
Furthermore, this CDC guidance would apply to adults at increased risk of exposure to poliovirus, including travelers who are going to countries where polio is epidemic or endemic.
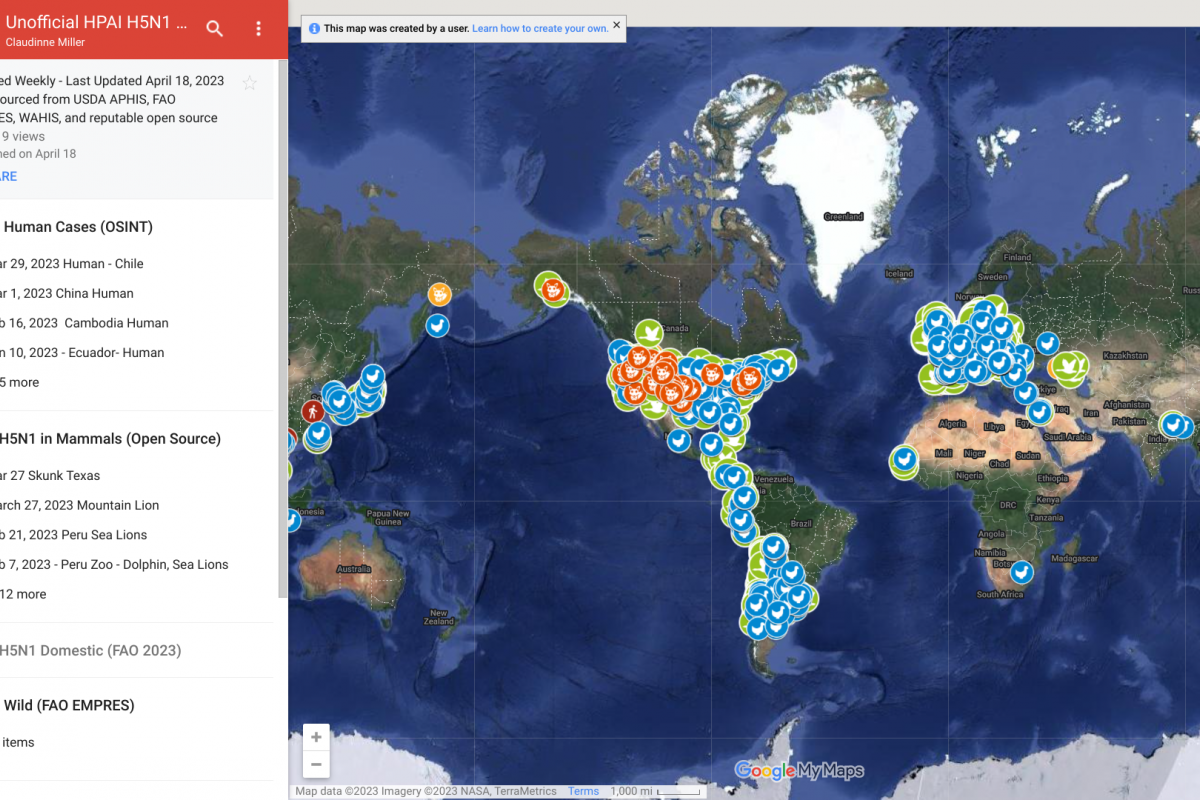
Globally, several countries have reported polio outbreaks in 2023. To alert international travelers, the CDC recently reissued a polio alert.