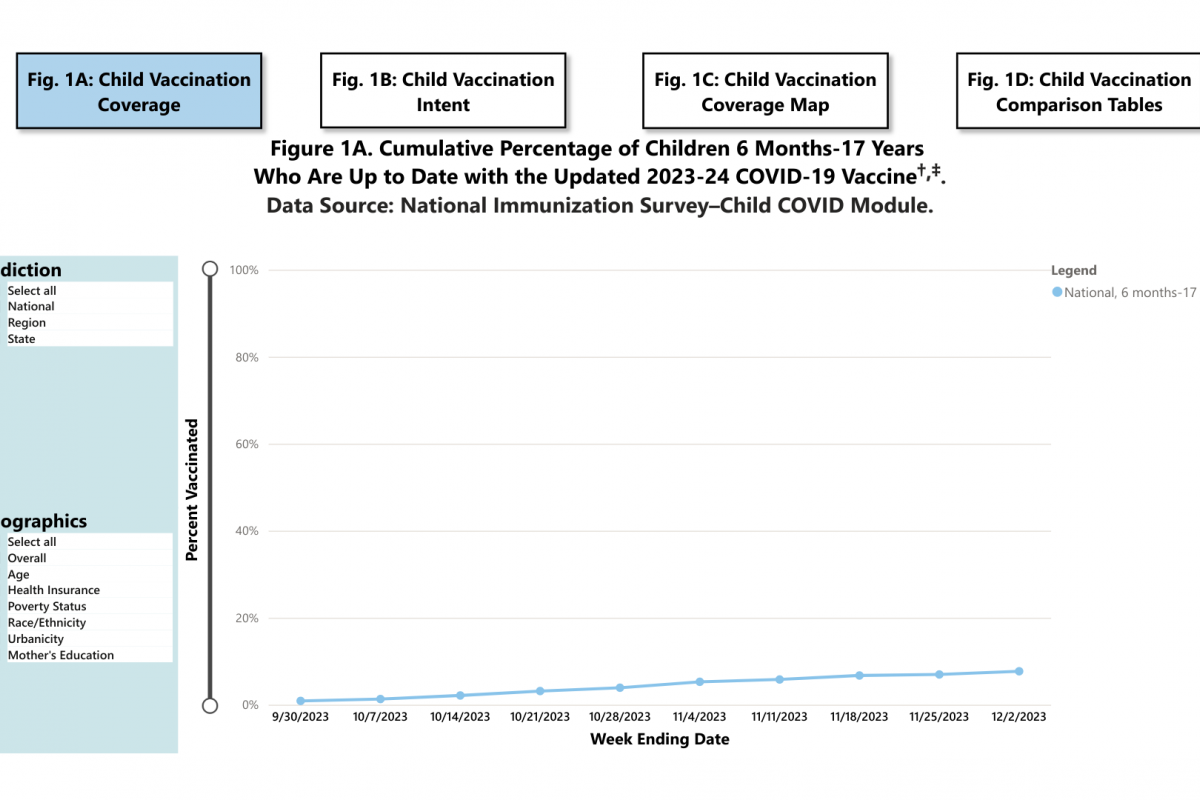
A Yale University-led study published today in the journal Pediatrics announced the rate of invasive pneumococcal disease (IPD) in children decreased by about 72% (incidence rate ratios 0.28, 95% CI 0.18–0.45) over twenty years.
This research found IPD rates continued to decline after the replacement of PCV7 with PCV13.
And during the recent pandemic, the rate of IPD among children aged <18 years reached 1.6 per 100,000, the lowest incidence observed over the 20 years.
In the PCV13 vaccine era, approximately one-third of the IPD cases in children aged >5 years had at least one underlying condition.
Serotypes 19A and 7F contributed 342 (48.9%) of all cases before implementation of PCV13 (2002–2010).
Serotype 3 (31, 8.6%), and non-PCV13 serotypes 15B/C (39, 10.8%), 33F (29, 8.0%), 23B (21, 0.8%), and 35B (17, 4.7%) were responsible for 37.8% of cases in PCV13 era (2011–2021).
Furthermore, penicillin nonsusceptibility declined (9.8% vs. 5.3% in the pre-/late PCV13 era, P = .003). However, it has become more common among non-PCV13 serotypes than vaccine serotypes (14.8% vs 1.4%, P < .001).
These Yale Child Health Research Center researchers concluded that robust ongoing surveillance networks are critical for identifying emerging pneumococcal serotypes and developing next-generation vaccine formulations.
As of December 2023, various pneumococcal vaccines are available at clinics and pharmacies in Asia, Europe, Japan, the U.S., and the United Kingdom.