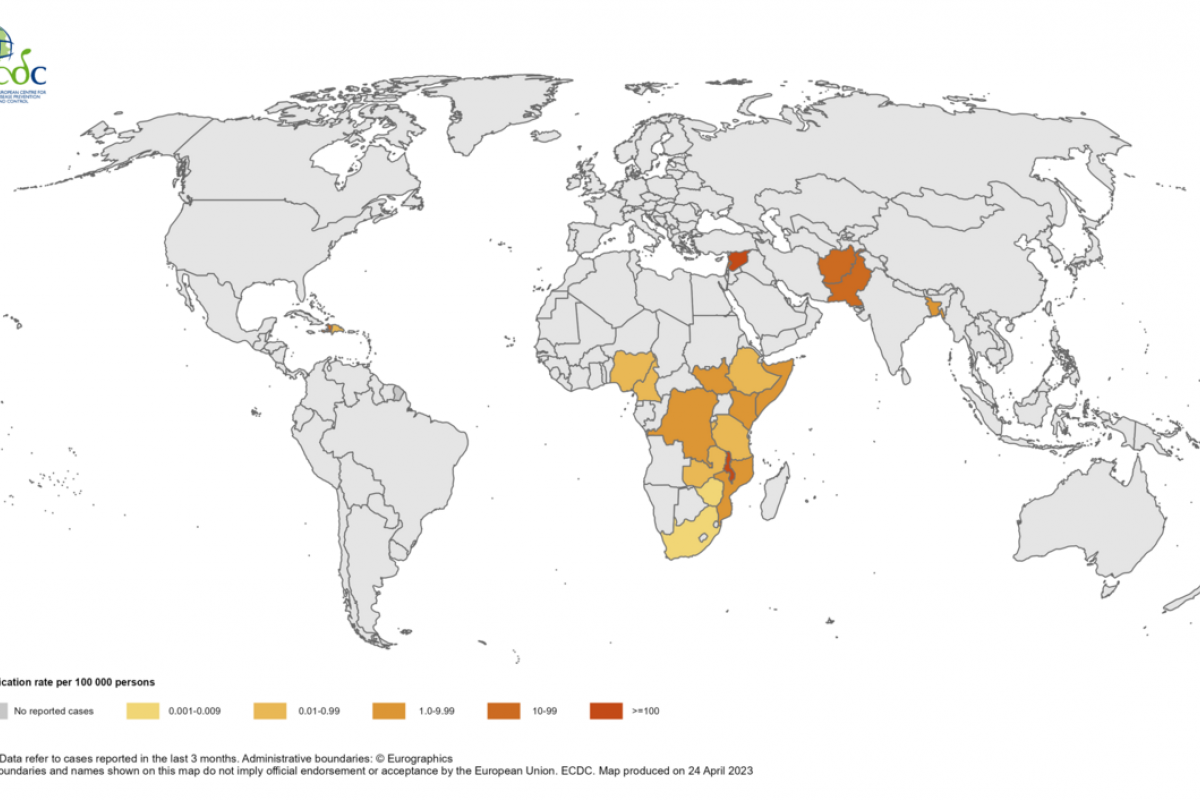
The Pan American Health Organization (PAHO) recently stated the detection of bird flu outbreaks in Latin America and the Caribbean is a situation never recorded before.
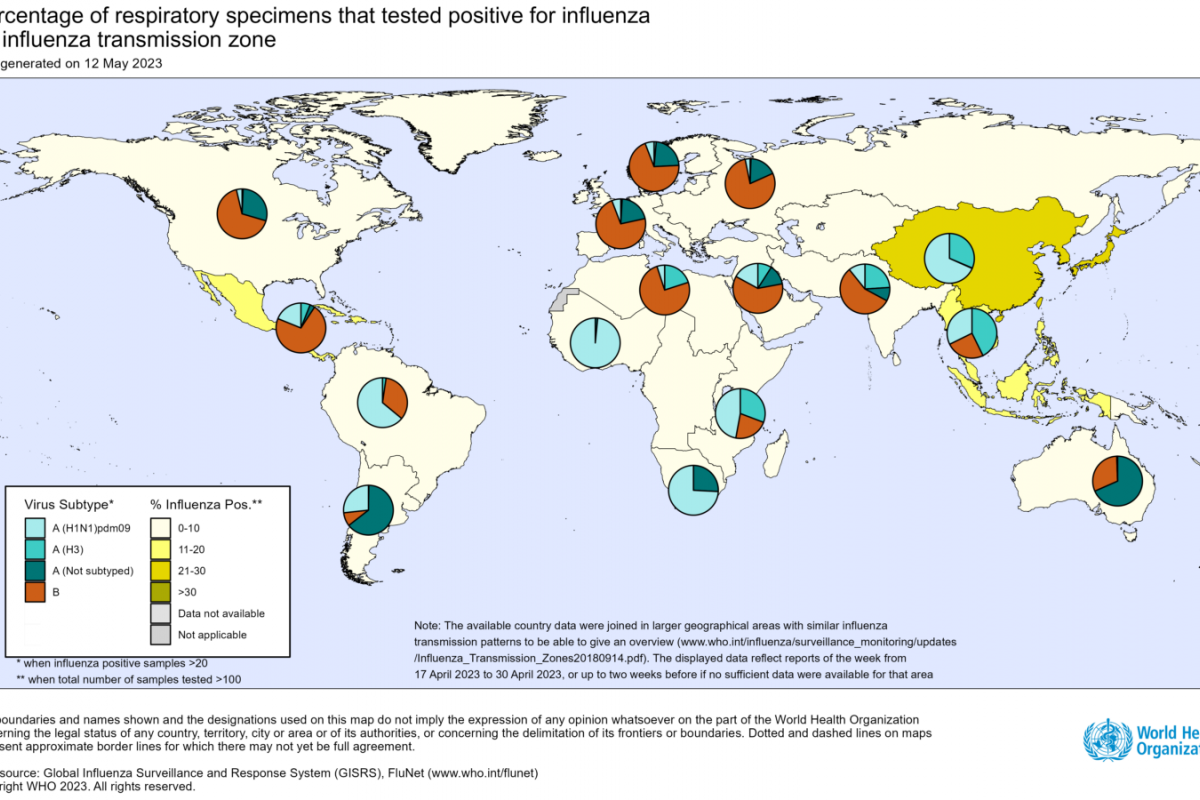
The identified influenza type A Highly Pathogenic Avian Influenza (HAPI) outbreaks are mainly located in areas of the Pacific flyway.
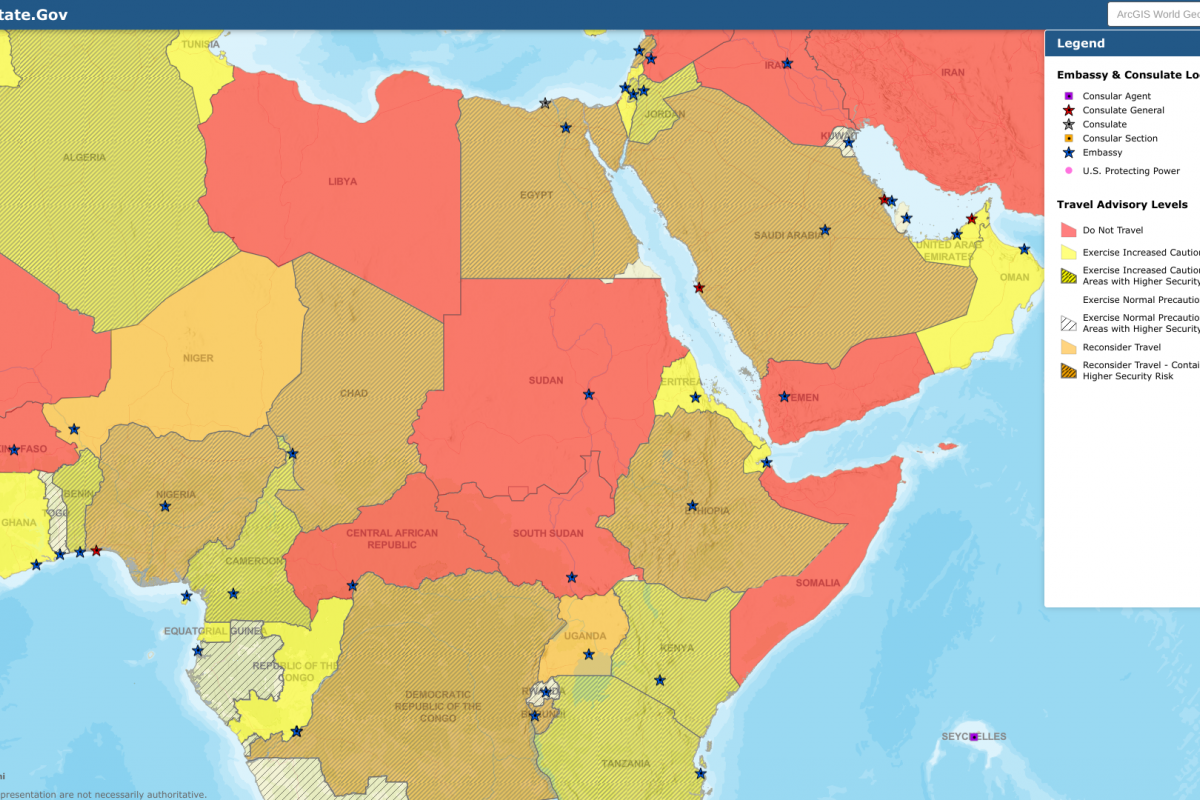
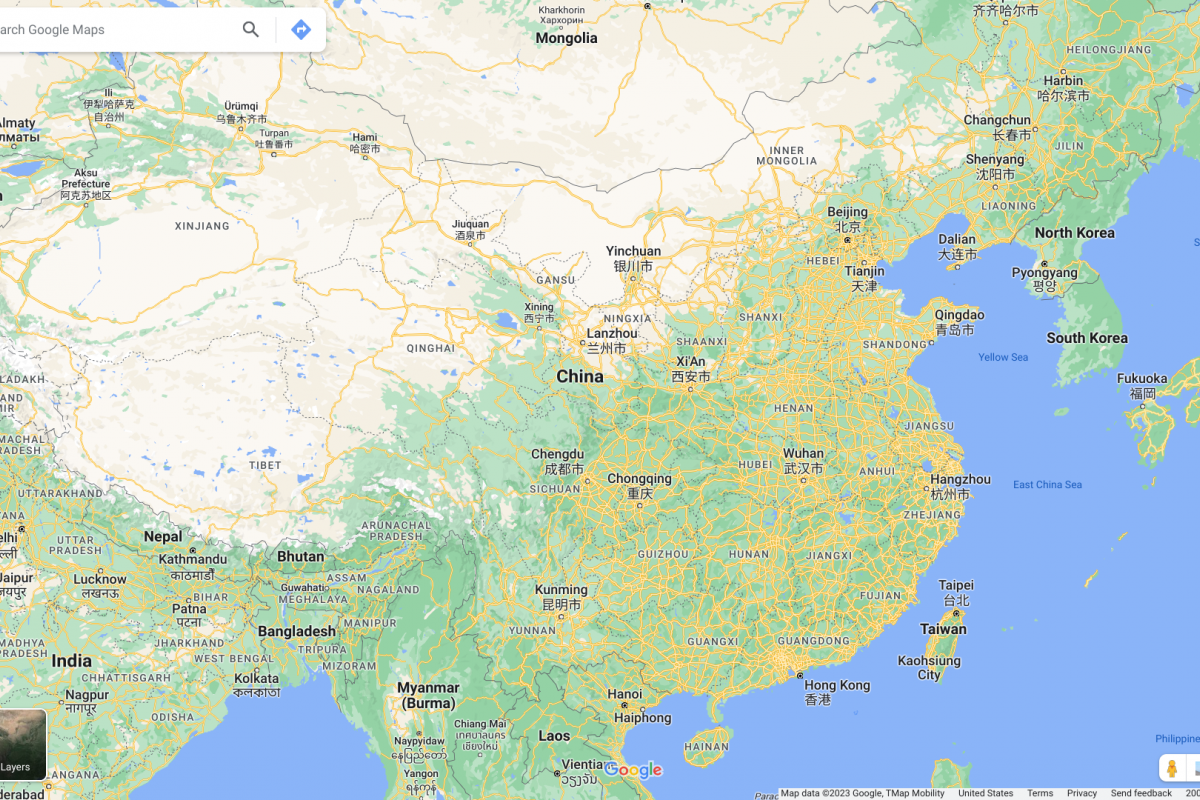
As of May 18, 2023, agricultural authorities in Argentina, Bolivia, Brazil, Canada, Chile, Colombia, Costa Rica, Cuba, Ecuador, Guatemala, Honduras, Mexico, Panama, Peru, U.S., Uruguay, and Venezuela have detected outbreaks of HPAI A(H5N1) viruses in domestic birds, farm poultry and/or wild birds, and in mammals.
Among the mammals identified, red foxes and skunks were the most frequently affected in North America, and fur seals in South America.
To date and since the introduction of avian influenza A (H5N1) in the Americas in 2014, three human infections caused by avian influenza A(H5N1) have been reported: the first in the U.S., reported in April 2022, the second in Ecuador, which was notified in January 2023, and the third in Chile, which was announced in March 2023.
In the U.S., the Audenz™ monovalent cell-based vaccine was authorized on January 31, 2020.
Recently, the U.S. FDA issued a Supplemental Approval in November 2021 for using Audenz in persons at increased risk of exposure to the influenza A virus H5N1 subtype.
The FDA confirmed annual flu shots are not designed to protect people from pandemic influenza.