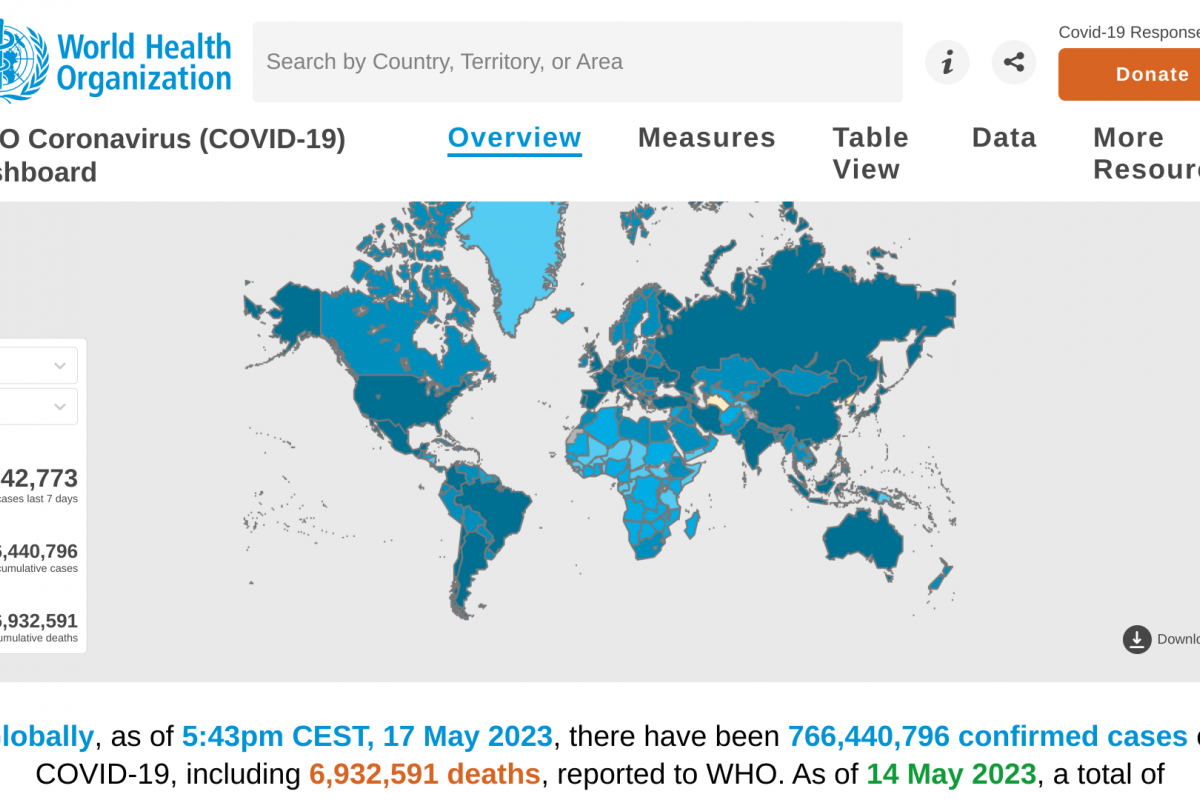
According to Paraguay's National Animal Health and Quality Service President, Dr. José Carlos Martin, on May 22, 2023, three confirmed outbreaks of avian influenza (bird flu) in the Chaco region occurred, while two others were still under investigation.
MercoPress, a news agency based in Uruguay, reported bird flu was registered in home-breeding establishments with open-air sheds and backyard poultry, in Mariscal Estigarribia, Colonia Neuland, and Filadelfia, in the department of Boquerón.
On May 18, 2023, the Pan American Health Organization announced bird flu outbreaks are mainly occurring in areas along the Pacific flyway and that outbreaks have occurred in 15 countries in Latin America and the Caribbean, which it said is unprecedented.
In North America, Canada and the United States have been battling the virus since early 2022 and have reported it in wild birds, poultry, and mammals.
Additional bird flu outbreak news regarding mammals and people is posted at Precision Vaccinations.